
If you've spent any time researching B vitamins for nerve health, you've probably encountered benfotiamine — usually with a claim somewhere in the description that it's “superior” to regular vitamin B1. That word “superior” made me skeptical the first time I saw it. Marketing claims about supplements being better than the standard version are everywhere, and most of the time they don't hold up to scrutiny.
Benfotiamine is different. There are actual structural and biochemical reasons why it behaves differently from ordinary thiamine in the body — reasons that have real implications for how well it reaches nerve tissue. That doesn't mean every claim made about it is accurate, and the clinical evidence for neuropathy specifically is more nuanced than many supplement websites let on. But it's worth taking seriously in a way that most “enhanced” supplement forms aren't.
Let's go through what benfotiamine actually is, what the science shows, who is most likely to benefit, and what dose the research used — so you can have an informed conversation with your doctor rather than just taking someone's word for it.
What Is Benfotiamine?
Benfotiamine is a synthetic derivative of thiamine — vitamin B1. It was developed in Japan in the 1950s, and by the 1990s it was being used in Germany and several other European countries as a prescription medication for diabetic neuropathy. It remains prescription-strength in parts of Europe while being available over the counter as a supplement in the United States.
What Makes Benfotiamine Different
Benfotiamine is lipid-soluble — it crosses gut and cell membranes through passive diffusion instead of a saturating transporter. This makes it approximately 5× more bioavailable than standard thiamine and better able to reach nerve tissue. The end result inside cells is the same active form of B1.
Structurally, the key difference from regular thiamine is that benfotiamine is lipid-soluble. Standard thiamine (thiamine hydrochloride, the form in most multivitamins) is water-soluble. That one difference has significant consequences for how the body absorbs and uses it.
Thiamine is essential for nerve function. It plays a critical role in glucose metabolism and in the production of ATP — the cellular energy currency. Nerve cells have extremely high energy demands, and they're particularly vulnerable to thiamine deficiency. This is why severe thiamine deficiency (as in alcoholic neuropathy or nutritional deficiency) causes one of the most dramatic forms of peripheral nerve damage: symptoms that can include burning pain, weakness, and sensory loss progressing from the feet upward.
For people whose neuropathy involves thiamine insufficiency — even subclinical insufficiency — getting more thiamine into nerve tissue makes intuitive biological sense. The question is whether benfotiamine actually gets there more effectively than standard thiamine, and whether that translates into meaningful clinical benefit.
How Benfotiamine Differs from Regular Thiamine
This is where the biochemistry actually supports the marketing claim, up to a point.
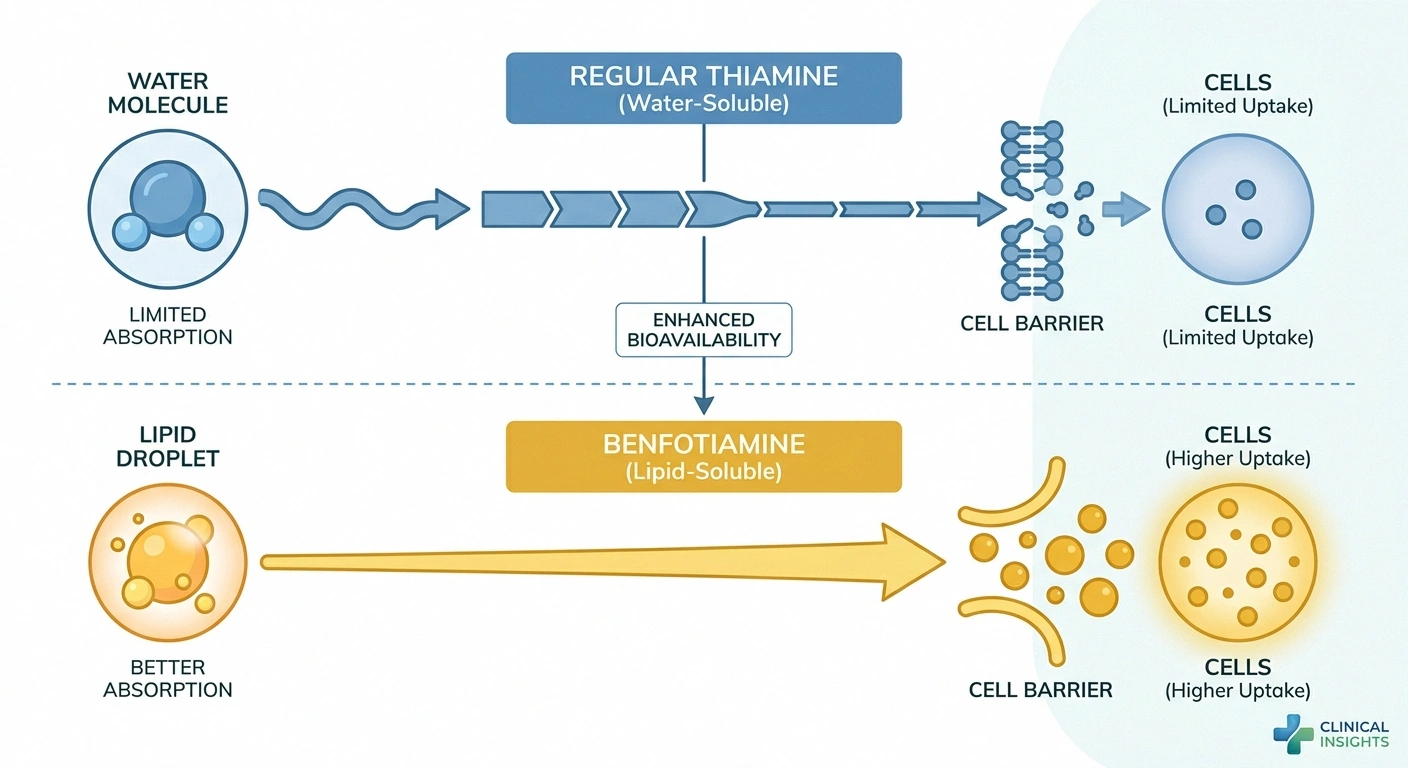
Regular thiamine is absorbed in the small intestine through an active transport system — a specific protein carrier that moves thiamine molecules across the gut wall. This system has a saturation ceiling. Once you take more thiamine than the transporter can handle, the excess isn't absorbed regardless of how large the dose is. This is why taking huge doses of standard thiamine supplements doesn't proportionally increase blood or tissue levels beyond a certain point.
Benfotiamine, being lipid-soluble, crosses the gut wall through passive diffusion — basically dissolving through the fatty membrane rather than waiting for a transporter. This bypasses the saturation ceiling. The result is substantially better bioavailability: research has consistently shown that benfotiamine raises thiamine levels in blood and tissues approximately five times more effectively than an equivalent oral dose of standard thiamine.
More importantly for neuropathy, benfotiamine reaches nerve tissue at meaningful concentrations. Standard thiamine, even in high doses, struggles to penetrate into nerve cells at levels that influence their function. Benfotiamine's lipid solubility allows it to cross biological membranes more readily — including the membranes of neurons and the myelin sheath that insulates nerve fibers.
Once inside cells, benfotiamine is converted to thiamine pyrophosphate, the biologically active form. From there, it functions exactly like thiamine — activating enzymes in glucose metabolism and supporting ATP production. The structural advantage is entirely in the delivery; the final action is the same as regular vitamin B1.
The Biochemical Mechanism: Four Pathways in Diabetic Nerve Damage
The most extensively researched application for benfotiamine is diabetic neuropathy, and the mechanism here is particularly compelling. Researchers have identified that high blood sugar damages nerves through at least four biochemical pathways:
How Benfotiamine May Protect Nerves in Diabetes
By activating the enzyme transketolase, benfotiamine redirects excess glucose away from 4 damaging pathways:
- The hexosamine pathway — converts glucose into compounds that damage proteins and cellular function
- Advanced glycation end-products (AGEs) — glucose molecules that stick to proteins and damage them structurally, including the proteins that make up nerve myelin
- The diacylglycerol-protein kinase C (DAG-PKC) pathway — disrupts blood vessel function and nerve blood supply
- NF-κB activation — triggers inflammatory gene expression that damages nerve cells
All four of these pathways are activated when glucose overwhelms the cell's normal processing capacity. Benfotiamine's mechanism of action is to activate an enzyme called transketolase, which reroutes excess glucose metabolites away from all four of these damaging pathways and into the safer pentose phosphate pathway.
The elegant thing about this mechanism is that it addresses the upstream problem — glucose toxicity to nerves — rather than just managing symptoms. If the mechanism holds up in human trials the way it does in laboratory models, benfotiamine could slow the progression of diabetic nerve damage, not just reduce pain. Whether that actually happens at the doses people can take safely is the question the clinical trials have been trying to answer.
What the Clinical Research Actually Shows
The honest answer here is: the evidence is promising but mixed, and it's weaker than the mechanism would predict. This is a pattern you see with many supplements — compelling biochemistry, disappointing or inconsistent trials.

Evidence Summary
Positive: 165-patient RCT showed significant symptom improvement at 600 mg/day over 6 weeks. Germany approved it as a prescription drug for diabetic neuropathy.
Mixed: 24-month RCT at 300 mg/day showed no improvement in nerve function. Dose may matter more than duration for achieving benefit.
The positive side:
A randomized controlled trial involving 165 patients with diabetic polyneuropathy found that taking 600 mg of benfotiamine per day for six weeks produced significantly better neuropathy symptom scores compared to placebo. This is a reasonably sized, properly controlled trial, and the result is meaningful. Patients reported improvements in pain, numbness, and tingling — the hallmark symptoms of diabetic neuropathy.
Earlier research, particularly a combination product (benfotiamine plus other B vitamins) used in Germany in the 1990s, showed improvements in neuropathic pain and vibration sensation in diabetic patients. Germany's decision to approve benfotiamine as a prescription treatment for diabetic neuropathy wasn't made on weak evidence.
For alcoholic neuropathy — where thiamine deficiency is often a direct contributor to the nerve damage — the case for thiamine supplementation (in any form) is strong, and benfotiamine's superior bioavailability makes it the logical choice over standard thiamine in this context.
The less encouraging side:
A well-designed 24-month randomized controlled trial published in a prominent diabetes journal followed patients with type 1 diabetes taking 300 mg of benfotiamine daily. The result: no significant effect on peripheral nerve function or inflammatory markers. This was a rigorous, long-term trial — and it came up negative at 300 mg/day.
This null result doesn't invalidate the positive studies, but it does complicate the picture. The difference between the positive six-week trial (600 mg/day) and the negative 24-month trial (300 mg/day) might come down to dose — 600 mg/day may be necessary for clinically relevant effects, while 300 mg/day may be insufficient. Or the benefit may be more pronounced in symptomatic short-term outcomes than in objective nerve function measures over time. Or the benefit may be specific to certain patient populations (type 2 diabetes rather than type 1, for example).
What the research does not support is the broader claim, made by some supplement marketers, that benfotiamine is a proven treatment for all forms of peripheral neuropathy. The research is specifically in diabetic and alcoholic neuropathy, where thiamine metabolism is directly relevant. For idiopathic neuropathy, autoimmune neuropathy, or neuropathy from other causes, the evidence base is thin to nonexistent.
Benfotiamine vs. Other B Vitamin Supplements for Neuropathy
Understanding where benfotiamine fits in the landscape of B vitamins for nerve health requires knowing what role each B vitamin plays.
B12 deficiency is probably the most common vitamin deficiency linked to neuropathy, and it's also one of the most clearly treatable. If you're B12 deficient — which is common in older adults, people on metformin, and those with absorption issues — supplementing B12 or getting B12 injections can genuinely improve neuropathy symptoms. B12 deficiency neuropathy and B1 deficiency neuropathy are different conditions, and supplementing B1 (as benfotiamine) will not address a B12 deficiency.
B6 (pyridoxine) is interesting because it's both potentially helpful for neuropathy in deficiency AND a cause of neuropathy in toxicity. Doses above 200 mg/day of B6 taken long-term can cause sensory neuropathy — a fact that's buried in many supplement marketing materials. If you're taking a high-dose B6 supplement, checking your levels with a simple blood test is worth doing.
Where does benfotiamine fit compared to these? If your neuropathy is specifically related to thiamine insufficiency (most likely in diabetic neuropathy or alcoholism), benfotiamine's superior bioavailability makes it substantially more useful than standard thiamine. For B12-deficiency neuropathy, benfotiamine is irrelevant. For neuropathy from other causes, neither B12 nor benfotiamine is the primary intervention — though they may be worth adding as supportive measures alongside primary treatment.
It's also worth knowing about a combination product called Milgamma (popular in Europe and used in some of the older German research), which combines benfotiamine with pyridoxine and cyanocobalamin (B12). The combination may address multiple B vitamin pathways simultaneously. Some researchers believe the combination outperforms benfotiamine alone, though head-to-head trial data is limited.
Dosage: What the Research Used
This is an area where being specific matters, because the doses used in published research vary significantly, and the over-the-counter products available often contain lower doses than what the trials used.
Dose at a Glance
- Positive six-week trial (diabetic polyneuropathy): 600 mg/day
- Negative 24-month trial (type 1 diabetes): 300 mg/day
- Prescription formulations in Europe: typically 300–600 mg/day in divided doses
- Common OTC supplements: typically 80–300 mg per capsule
The takeaway is that many over-the-counter benfotiamine products are at the low end of the studied dose range or below it. If you're going to try benfotiamine seriously, using a dose of at least 300 mg/day and possibly up to 600 mg/day is more consistent with the positive research — but this is a conversation to have with your doctor, particularly because benfotiamine is sometimes used alongside other supplements and medications that can interact.
Benfotiamine can be taken with or without food. Some sources suggest dividing the daily dose into two portions (morning and evening) for more consistent blood levels throughout the day, though there's no definitive research showing this is necessary.
Side Effects and Safety
The safety profile for benfotiamine is generally favorable. Clinical trials have not identified serious adverse events at therapeutic doses. The most commonly reported side effects, when they occur, are mild gastrointestinal symptoms — nausea, upset stomach, or loose stools — and occasional skin reactions.
Unlike vitamin B6 at high doses, there's no known toxicity syndrome for benfotiamine or thiamine generally. The body excretes excess water-soluble B vitamins (regular thiamine), and while benfotiamine's better tissue penetration means it stays in the body somewhat longer, there's no evidence of accumulation to dangerous levels at the doses used in research.
The usual caveats apply: if you're pregnant, breastfeeding, have a significant kidney condition, or are taking blood-thinning medications, check with your doctor before adding any new supplement. There's limited specific safety data for benfotiamine in these populations.
Who Is Most Likely to Benefit?
Based on the existing research, benfotiamine is most likely to help:

Most Likely to Benefit from Benfotiamine
- People with diabetic peripheral neuropathy (strongest evidence)
- People with alcoholic neuropathy (thiamine deficiency component)
- Those with subclinical thiamine insufficiency (poor diet, IBD, bariatric surgery)
- People on metformin (depletes B12 and thiamine over time)
Less likely to help: autoimmune neuropathy, mechanical compression neuropathy, or neuropathy from causes unrelated to metabolic or nutritional factors.
People with diabetic neuropathy — This is where the mechanism is strongest and the most clinical evidence exists. If you have diabetic neuropathy, benfotiamine addresses the glucose-toxicity pathways that are directly damaging your nerves. The symptomatic benefit in the positive trial was meaningful, and given the safety profile, the risk-benefit calculation is generally favorable as an add-on to standard diabetic neuropathy management.
People with alcoholic neuropathy — Thiamine deficiency is a direct component of alcoholic neuropathy. Benfotiamine's superior tissue penetration makes it the logical thiamine formulation for addressing this deficiency compared to standard thiamine supplements.
People with borderline or subclinical thiamine insufficiency — This can occur in malnutrition, inflammatory bowel disease, bariatric surgery, chronic poor diet, and certain medications. Even without overt deficiency, getting more thiamine into nerve tissue may be supportive.
People with general peripheral neuropathy looking for adjunctive support — The evidence is weaker here, but given the safety profile, benfotiamine as part of a comprehensive neuropathy supplement protocol alongside alpha-lipoic acid and B12 is a reasonable approach for many patients.
Benfotiamine is less likely to help if your neuropathy is primarily driven by autoimmune damage, mechanical compression, or causes unrelated to metabolic or nutritional pathways. It's also not a substitute for controlling blood sugar in diabetic neuropathy — it works on the consequences of hyperglycemia, not on the underlying blood sugar problem itself.
How Long Before You Might See Results?
The positive six-week trial showed measurable improvement in symptom scores within six weeks. That's a reasonable initial trial period — if you're going to try benfotiamine, give it at least 6–8 weeks at an appropriate dose before deciding whether it's helping.

Some patients report improvements in burning and tingling within two to four weeks. Others notice nothing for months and then gradual improvement. Nerve healing is slow — peripheral nerves regenerate at a rate of approximately 1 mm per day, meaning structural repair takes months to years. Symptom improvement can precede structural improvement if benfotiamine is reducing ongoing damage and reducing pain signals even before nerves fully recover.
It's worth tracking your symptoms with a simple symptom log when starting any new supplement for neuropathy. Neuropathy symptoms fluctuate naturally — they're often worse in cold weather, at night, when fatigued, or during stress. Without a baseline and consistent tracking, it's genuinely hard to tell whether a supplement is helping or whether you just happened to have a good week.
The complete picture of natural remedies for neuropathy involves multiple approaches working together — diet, alpha-lipoic acid, B vitamins including benfotiamine where appropriate, exercise, and blood sugar management for diabetic patients. No single supplement, including benfotiamine, is likely to fully resolve established neuropathy on its own. But as part of a thoughtful, multi-component approach, the evidence suggests it can meaningfully contribute for the right patients.
Frequently Asked Questions
Is benfotiamine the same as vitamin B1?
Benfotiamine is a synthetic derivative of vitamin B1, also called thiamine. It produces the same biologically active form inside cells as regular thiamine, but its lipid-soluble structure allows it to be absorbed significantly better through the gut and to penetrate into tissues, including nerve cells, more effectively than water-soluble standard thiamine. It is not identical to the thiamine you get from food or from most multivitamins, but it ends up doing the same job once inside cells. Bioavailability studies show benfotiamine achieves approximately five times higher tissue thiamine levels compared to an equivalent oral dose of standard thiamine.
Does benfotiamine help all types of neuropathy?
The clinical evidence is strongest specifically for diabetic neuropathy and alcoholic neuropathy, where thiamine metabolism plays a direct role in nerve damage. For other forms of neuropathy, including idiopathic neuropathy, autoimmune neuropathy, and hereditary neuropathy, there is little specific research. Benfotiamine may still be worth considering as a supportive measure alongside primary treatments for other neuropathy types, given its safety profile and the fundamental role of thiamine in nerve function, but patients with non-diabetic, non-alcoholic neuropathy should have realistic expectations about the evidence base.
What dose of benfotiamine should I take for neuropathy?
The research showing benefit for diabetic neuropathy used 600 mg per day. The research showing no benefit used 300 mg per day over two years. Common over-the-counter products contain 150 to 300 mg per capsule. The evidence suggests that doses at or near 600 mg per day may be necessary for meaningful symptomatic benefit, but this should be discussed with a healthcare provider. Your specific neuropathy type, other medications, and overall health picture should factor into the dosing decision. Do not exceed recommended doses without medical guidance.
Is benfotiamine safe to take long-term?
Clinical trials lasting up to two years have not identified serious safety concerns with benfotiamine at therapeutic doses. There is no known toxicity syndrome associated with excess thiamine or benfotiamine the way there is with certain other vitamins. Mild gastrointestinal side effects are the most commonly reported issue and tend to be temporary. The safety profile is generally favorable compared to most pharmaceutical neuropathy medications. As with any supplement taken long-term, periodic review with a healthcare provider is sensible.
Can benfotiamine be taken with other neuropathy supplements?
Benfotiamine is commonly combined with other B vitamins, particularly B12 and B6, and with alpha-lipoic acid for neuropathy support. European prescription products (such as Milgamma) specifically combine benfotiamine with B12 and B6, and there is research suggesting the combination may outperform benfotiamine alone. Be careful with high-dose B6 supplementation, as doses above 200 mg per day taken long-term can cause nerve damage. Tell your doctor about all supplements you are taking, particularly if you are also on medications that affect B vitamin metabolism such as metformin.
How is benfotiamine different from the benfotiamine in prescription medications?
In countries like Germany where benfotiamine is approved as a prescription drug for diabetic neuropathy, the prescription product is the same compound as the over-the-counter supplement sold in the United States. The main differences are the dose certainty, manufacturing quality standards, and the regulatory oversight that comes with prescription status. Over-the-counter supplements in the US are not held to the same quality standards as pharmaceutical products, which means the actual benfotiamine content may vary. If you are purchasing benfotiamine as a supplement, look for products from manufacturers who publish third-party testing results.
Should I take benfotiamine if I also take metformin for diabetes?
This is a particularly relevant question because metformin is known to deplete both vitamin B12 and thiamine over time, and both deficiencies can contribute to peripheral neuropathy. People taking metformin long-term are at elevated risk for B12 deficiency neuropathy specifically, and benfotiamine may provide additional thiamine-related support. However, B12 deficiency neuropathy requires B12 supplementation directly, not thiamine. Anyone on metformin with neuropathy symptoms should have both B12 and thiamine status assessed. Your doctor can advise on whether benfotiamine is appropriate alongside any B12 supplementation already in your regimen.


