When my neurologist first told me that some of my nerve fibers had lost their “insulation,” I nodded like I understood. I didn't, really. I just knew something was damaged and that it hurt.
It took me a while to actually learn what myelin is and why losing it matters so much. Once I did, a lot of things clicked into place — why my symptoms felt the way they did, why some types of neuropathy progress faster than others, and why certain treatments work for some people but not others.
If you're living with neuropathy or caring for someone who is, understanding the myelin sheath isn't just biology trivia. It's the foundation for understanding what's actually happening in your nervous system and what that means for your treatment options and long-term outlook.
What Is the Myelin Sheath?
Imagine a nerve fiber as an electrical wire. The wire itself (the axon) carries the signal. The insulation wrapping that wire is the myelin sheath — a fatty, protein-rich coating that surrounds the axon and plays several critical roles.
Key Takeaway
The myelin sheath is the fatty insulation around nerve fibers that makes signals travel up to 60x faster. When it's damaged (demyelinating neuropathy), signals slow or fail — causing weakness, numbness, and pain. Unlike axonal damage, peripheral myelin can regenerate if the underlying cause is stopped.
Myelin isn't applied continuously like paint. It's wrapped in segments, with small gaps between each segment called nodes of Ranvier. These gaps are strategically placed and critically important for how nerves actually work.
Myelin is produced by specialized cells:
- In the peripheral nervous system (the nerves outside your brain and spinal cord), myelin is made by Schwann cells
- In the central nervous system (brain and spinal cord), myelin is made by oligodendrocytes
This distinction matters for understanding why peripheral neuropathy and central nervous system conditions like multiple sclerosis are different diseases with different behaviors, even though both involve myelin damage.
What Does Myelin Actually Do?
Myelin serves three fundamental functions in the nervous system:
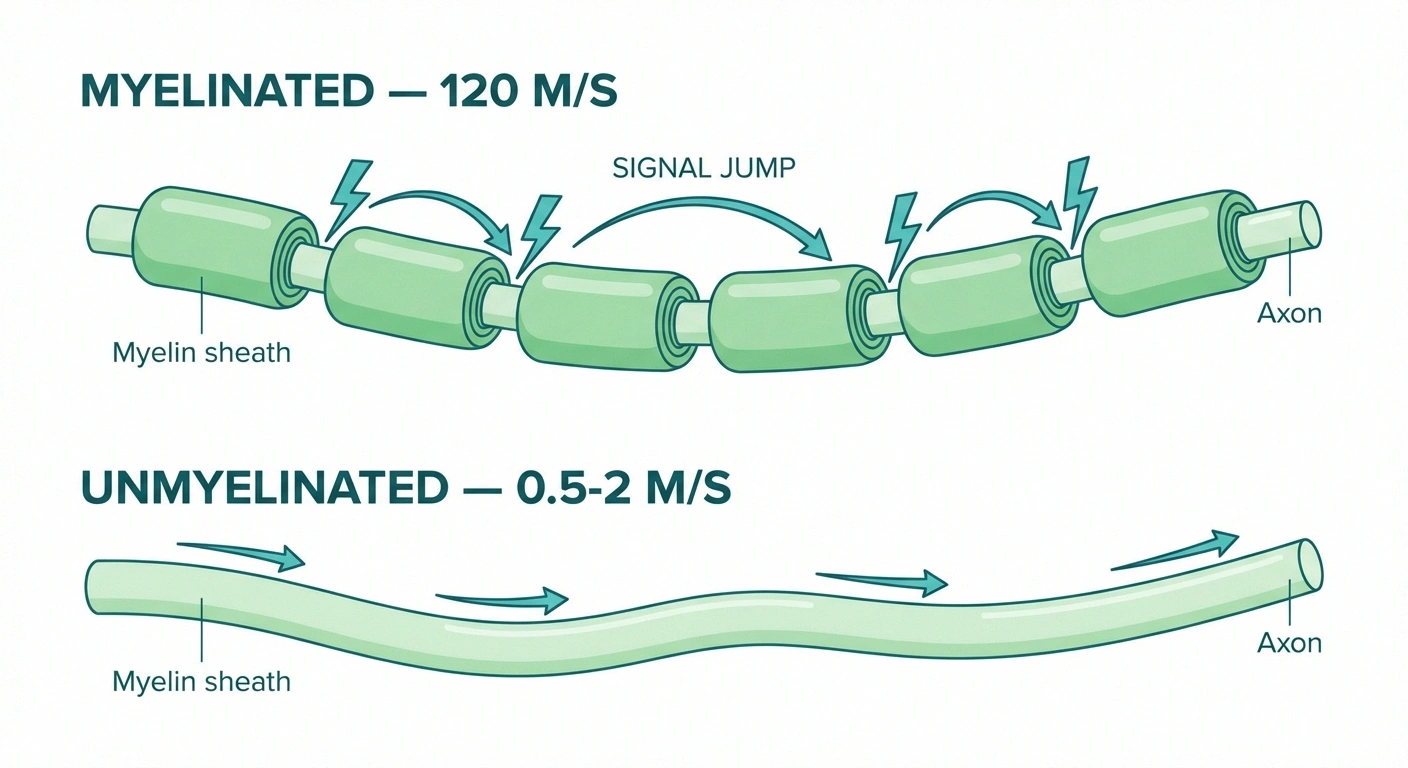
Nerve Signal Speed: Myelinated vs. Unmyelinated
120 m/s
Myelinated (A-alpha)
Signal jumps between nodes
1. Massively Speeds Up Signal Transmission
Without myelin, electrical impulses travel along the nerve fiber by continuously exciting each part of the membrane in sequence — like a slow burning fuse. This is called continuous conduction, and it's slow: typically 0.5 to 2 meters per second.
With myelin, the electrical signal essentially jumps from one node of Ranvier to the next, skipping across the insulated sections entirely. This is called saltatory conduction (from the Latin “saltare,” to jump), and it's dramatically faster — up to 120 meters per second in the most heavily myelinated fibers. That's the difference between a slow creep and nearly instantaneous transmission.
For context: the reason you can catch something before it hits the floor, or recoil from a hot surface in a fraction of a second, depends on this extraordinary signal speed. When myelin is lost, these reactions slow down.
2. Improves Energy Efficiency
Continuous conduction requires ion pumping along the entire length of the nerve fiber — an energy-expensive process. Saltatory conduction concentrates this work at the nodes of Ranvier only, making myelinated fibers roughly 100 times more energy-efficient than unmyelinated ones. For a nervous system with billions of active nerve fibers, this efficiency is essential.
3. Protects and Supports the Axon
Myelin isn't just insulation — it provides metabolic support to the axon beneath it. Schwann cells supply nutrients and maintain the health of the nerve fiber they wrap. When myelin is damaged or destroyed, the axon it protects becomes vulnerable. In severe demyelination, the axon itself eventually degenerates — a process that is more permanent than myelin loss alone.
Myelinated vs. Unmyelinated Nerve Fibers: Why Both Matter for Neuropathy
Not all nerve fibers are myelinated — and which fibers are affected tells you a lot about what kind of neuropathy someone has.
Heavily myelinated fibers (A-alpha and A-beta): Carry motor signals and touch/pressure information. Very fast. When damaged, produce impaired coordination, muscle weakness, and loss of the ability to feel light touch or vibration.
Lightly myelinated fibers (A-delta): Carry sharp, fast pain signals and some temperature information. Damage produces altered pain sensing and temperature perception.
Unmyelinated fibers (C-fibers): Carry slow, burning pain, temperature, and autonomic signals. These are the fibers targeted in small fiber neuropathy — and because they're not captured by standard EMG/nerve conduction studies (which only measure the fastest myelinated fibers), small fiber neuropathy is often missed unless a skin punch biopsy is performed.
This fiber type differentiation explains why two people can both have “neuropathy” but have completely different symptoms: one may experience primarily burning and temperature dysregulation (small fiber), while another experiences more weakness, coordination problems, and vibration loss (large fiber demyelinating).
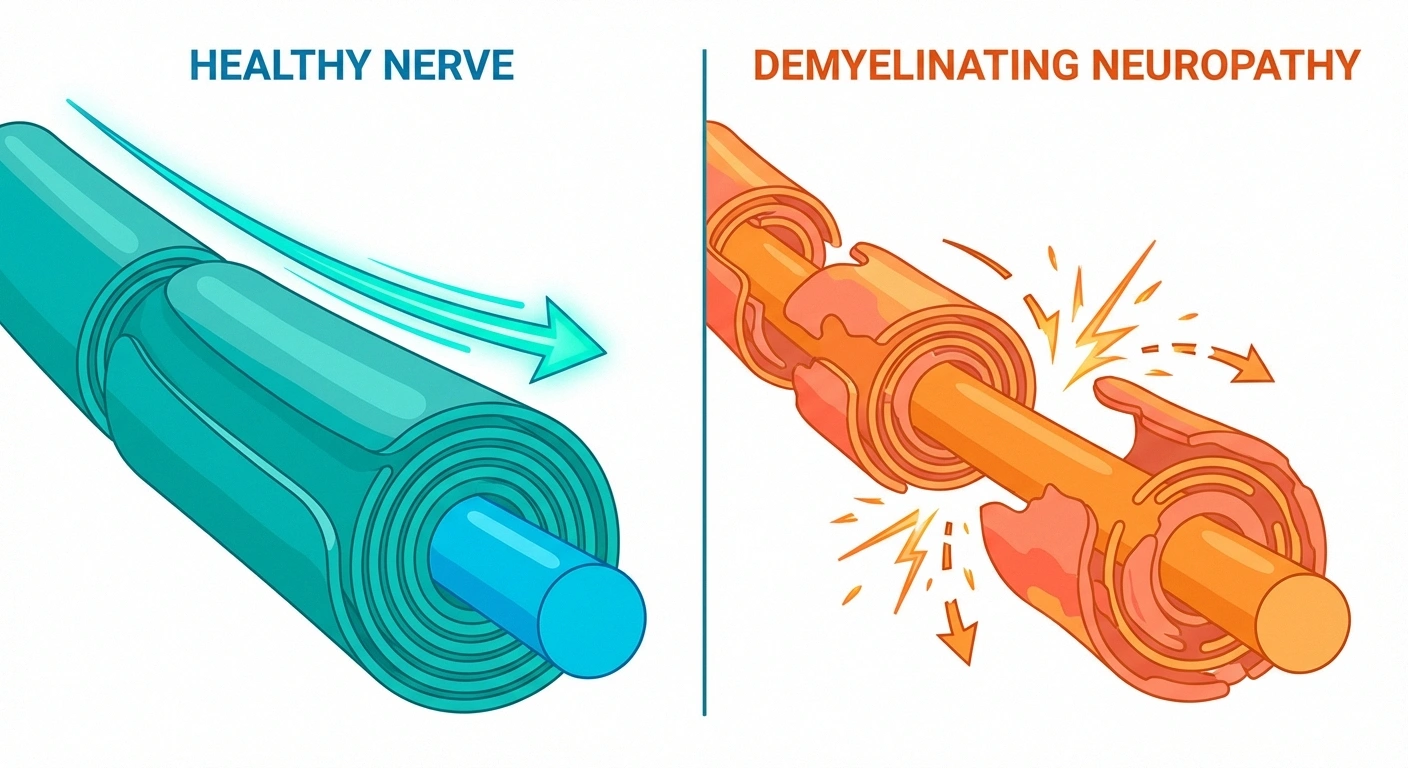
What Is Demyelinating Neuropathy?
Demyelinating neuropathy refers specifically to peripheral neuropathy where myelin damage is the primary pathology — as opposed to axonal neuropathy, where the axon itself is the primary target of damage.
Demyelinating vs. Axonal Neuropathy: Key Differences
| Feature | Demyelinating | Axonal |
|---|---|---|
| What's damaged | Myelin sheath | Axon itself |
| NCS finding | Slow velocity, preserved amplitude | Reduced amplitude, near-normal velocity |
| Recovery potential | Good (Schwann cells can remyelinate) | Slower (axon regenerates 1-3mm/day) |
| Common examples | GBS, CIDP, CMT Type 1 | Diabetic, alcoholic, toxic neuropathy |
| Responds to immunotherapy? | Often yes (if immune-mediated) | Generally no |
In demyelinating neuropathy:
- Signal conduction slows dramatically because saltatory transmission is disrupted
- The axon initially remains relatively intact, which means there's potential for recovery if the myelin can be restored
- Nerve conduction studies typically show slowed conduction velocity and prolonged latencies — the electrical signature of myelin damage
- Deep tendon reflexes (like the knee jerk) are often absent or reduced, because the reflex arc requires fast-conducting myelinated fibers
In axonal neuropathy, by contrast, the nerve fiber itself degenerates, amplitude on nerve conduction studies drops, and recovery is generally slower and less complete because axonal regeneration is a much slower process than remyelination.
Major Demyelinating Neuropathies
Guillain-Barré Syndrome (GBS)
GBS is an acute, immune-mediated attack on peripheral nerve myelin — usually triggered by a preceding infection. The immune system, in fighting off the infection, mistakenly attacks the myelin coating of peripheral nerves. GBS typically progresses over days to weeks, causing ascending weakness and sensory changes that can, in severe cases, lead to respiratory failure.
The important thing about GBS is that because the axons initially remain relatively intact, most patients recover significantly with treatment (IVIG or plasma exchange) and time. Recovery happens through remyelination — Schwann cells regenerate the myelin sheath over a period of weeks to months. Full recovery is common, though the timeline varies.
CIDP (Chronic Inflammatory Demyelinating Polyneuropathy)
CIDP is like GBS's chronic sibling — the same immune-mediated myelin attack, but persisting or relapsing rather than acute. CIDP progresses more slowly, typically over months, and requires ongoing treatment to suppress the immune attack and allow remyelination. IVIG, corticosteroids, and plasma exchange are the primary treatments. Response to these therapies confirms the demyelinating, immune-mediated nature of the condition.
Hereditary Demyelinating Neuropathies (CMT)
Charcot-Marie-Tooth disease (CMT) encompasses a group of inherited neuropathies, many of which are demyelinating. In CMT Type 1 (the most common form), mutations affect genes involved in myelin production or maintenance — particularly PMP22, which codes for a protein that's a structural component of the peripheral nerve myelin sheath. The resulting myelin is abnormal, leading to progressive demyelination and the characteristic presentation of CMT: slowly progressive distal weakness, foot deformities, and sensory loss.
Multifocal Motor Neuropathy (MMN)
MMN is a rare immune-mediated neuropathy characterized by asymmetric motor weakness without sensory loss, caused by conduction block in motor nerve fibers — a specific form of demyelination where the signal fails to pass a blocked segment. It's important to diagnose because it's treatable (with IVIG) and can be mistaken for motor neuron disease.
Can Myelin Regenerate?
This is the question everyone wants answered. The encouraging answer is yes — peripheral nerve myelin can regenerate. Schwann cells retain the ability to re-wrap demyelinated axons throughout life, unlike in the central nervous system where remyelination is more limited.
Research Says — 2025
Researchers at the University of Colorado Anschutz identified an experimental drug that accelerates remyelination and restored visual function in mice with demyelinating optic nerve damage. While still in early stages, this research points toward future therapies that could actively speed peripheral remyelination — a field of growing scientific investment.
In peripheral demyelinating neuropathy:
- If the underlying cause of demyelination is removed or suppressed (immune attack stopped, toxin removed, nutritional deficiency corrected), Schwann cells begin rebuilding the myelin sheath
- The regenerated myelin is initially thinner than the original, with shorter internodal segments — this is why nerve conduction remains somewhat slower even after recovery than before the damage occurred
- In GBS, visible nerve conduction improvement on follow-up studies corresponds to the remyelination process, and typically parallels clinical improvement
However, there are important limits to this regenerative potential:
- Ongoing demyelination prevents recovery — remyelination requires the causative attack to be stopped or slowed
- If demyelination is severe enough and prolonged enough, axonal degeneration follows — and axonal regeneration is slower (1-3mm per day) and less complete than remyelination
- Age affects regenerative capacity — repair proceeds more slowly in older individuals
Research in this area is progressing rapidly. A 2025 study from the University of Colorado identified an experimental drug that accelerates remyelination in animal models and restored visual function in mice with optic nerve demyelination — pointing toward the possibility of medications that could actively speed the remyelination process. This research currently focuses on central nervous system myelin, but the principles are informing peripheral nervous system research as well.
How Myelin Damage Creates Neuropathy Symptoms
Understanding the myelin sheath helps explain the specific character of demyelinating neuropathy symptoms, which differ somewhat from axonal neuropathy.
Slowed and Unreliable Conduction
When myelin is patchy or absent, nerve conduction slows and becomes variable. Mild demyelination may produce little more than subtle slowing that shows on tests but causes few symptoms. More significant demyelination begins causing symptoms: numbness and tingling from sensory fibers conducting erratically, weakness from motor fibers not reliably transmitting signals to muscles, and balance problems from proprioceptive fibers losing their fast-conduction accuracy.
Conduction Block
In some forms of demyelinating neuropathy, signals don't just slow — they fail entirely at focal areas of severe damage. This conduction block produces sudden loss of motor or sensory function in the distribution of the affected nerve — potentially explaining why some demyelinating neuropathy symptoms can appear quite suddenly.
Ectopic Discharge
Partially demyelinated nerve segments can also fire spontaneously — generating false signals that the brain interprets as pain, tingling, or other sensations. This mechanism contributes to the positive symptoms (things you feel, rather than things you can't feel) of demyelinating neuropathy: burning sensations, electric shocks, tingling.
Diagnosing Demyelinating vs. Axonal Neuropathy
The distinction between demyelinating and axonal neuropathy is important for diagnosis and treatment, and nerve conduction studies are the primary tool for making this distinction.

In a nerve conduction study, electrodes measure the speed and amplitude of nerve signals. The key findings:
- Demyelinating pattern: Slowed conduction velocity, prolonged distal latency, prolonged F-wave latency. Amplitude is relatively preserved (because the axon is intact). Conduction block may be present.
- Axonal pattern: Reduced amplitude (fewer functional axons). Conduction velocity relatively preserved (the remaining axons, which have intact myelin, conduct at normal speed).
- Mixed pattern: Both slow velocity and reduced amplitude — indicates damage to both myelin and axons.
The neuropathy diagnosis process typically combines nerve conduction studies with clinical examination and blood tests to determine both the pattern (demyelinating vs. axonal) and the likely cause.
What Causes Myelin Damage in Peripheral Neuropathy?
Understanding the cause of myelin damage is essential because the treatment follows from the cause.

What Causes Myelin Damage in Peripheral Neuropathy?
Autoimmune Attack
GBS, CIDP — immune system targets Schwann cells or myelin proteins
Autoimmune Attack
The immune system mistakenly targets myelin or Schwann cells. This is the mechanism in GBS, CIDP, and some cases of neuropathy associated with Sjögren's syndrome, lupus, and other connective tissue diseases. Treatment focuses on immunosuppression or immunomodulation — stopping the attack to allow remyelination.
Genetic Abnormalities
Mutations in genes that code for myelin proteins or Schwann cell function lead to abnormal myelin formation from birth. CMT is the most common example, with over 80 gene variants identified. There's no treatment that corrects the underlying genetic cause, though research into gene therapy is advancing.
Metabolic and Nutritional Deficiencies
Vitamin B12 is essential for myelin maintenance. Deficiency causes a characteristic pattern of demyelination affecting both peripheral nerves (producing neuropathy) and the spinal cord (producing a condition called subacute combined degeneration). Correcting B12 deficiency can allow remyelination and symptom improvement, though severe or long-standing deficiency may cause irreversible axonal damage. Copper deficiency and vitamin E deficiency can also damage myelin.
Toxins and Medications
Some drugs and chemical exposures selectively damage Schwann cells or the myelin they produce. Heavy metals, some chemotherapy agents, and certain industrial chemicals can cause demyelinating neuropathy. Removing the exposure (when possible) is the critical first step in allowing recovery.
Diabetes
Interestingly, diabetic neuropathy is primarily axonal — chronic high blood sugar damages the axon and the small blood vessels that supply it. However, some demyelinating changes also occur in diabetic neuropathy, making it a mixed picture in many people.
What the Myelin Sheath Means for Your Treatment
Knowing whether your neuropathy is primarily demyelinating or axonal has real treatment implications:
- Immune-mediated demyelinating neuropathy (GBS, CIDP) responds to immunotherapy — IVIG, plasma exchange, steroids. This is meaningful and often dramatic treatment that can halt progression and allow significant recovery.
- Hereditary demyelinating neuropathy (CMT) doesn't have disease-modifying treatment currently, but physical therapy, orthotics, and symptom management remain important.
- Nutritional deficiency demyelination responds to replacing the missing nutrient — sometimes surprisingly quickly for symptoms of short duration.
- Axonal neuropathy generally doesn't respond to immunotherapy (because the axon, not myelin, is the target), and recovery is slower because axons regenerate much more slowly than myelin reforms.
This is why the effort to distinguish demyelinating from axonal on nerve conduction studies isn't just academic — it changes what your neurologist will recommend and what you might reasonably expect from treatment.
The Peripheral vs. Central Distinction: Why It Matters
People sometimes confuse peripheral demyelinating neuropathy with central demyelinating diseases like multiple sclerosis. The key distinction: MS attacks myelin in the central nervous system (brain and spinal cord), while peripheral demyelinating neuropathy attacks myelin in the peripheral nervous system.
The clinical presentations differ: MS typically produces relapsing-remitting neurological symptoms with a characteristic pattern on brain MRI, while peripheral demyelinating neuropathy produces symptoms that follow peripheral nerve distributions (hands and feet, ascending rather than patchy). The treatments are also entirely different.
While they're distinct conditions, there are cases where both peripheral neuropathy and CNS demyelination occur in the same person — particularly in some autoimmune conditions. This is another reason a thorough neurological evaluation matters when the picture isn't straightforward.
Protecting Your Remaining Myelin
Regardless of the cause or type of your neuropathy, supporting nerve health generally helps preserve whatever myelin remains and may support the remyelination process.

- B vitamin optimization: B12, B1 (thiamine), and B6 (at appropriate doses — excess B6 is itself toxic to nerves) all support nerve health and myelin maintenance. Have deficiencies tested and addressed.
- Blood sugar control: Even in non-diabetics, blood sugar spikes cause oxidative stress that damages nerve tissue and impairs Schwann cell function.
- Anti-inflammatory diet: Chronic inflammation is directly damaging to Schwann cells. An anti-inflammatory diet reduces this ongoing damage.
- Avoid known neurotoxins: Alcohol is directly toxic to Schwann cells. Smoking reduces blood flow and increases oxidative stress. Both accelerate nerve damage.
- Exercise: Physical activity increases nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF) — growth factors that support nerve and Schwann cell health.
None of these measures repair severely damaged myelin on their own, but they create the physiological conditions most conducive to whatever repair is possible — and they reduce the rate of ongoing damage.
Frequently Asked Questions
What is the myelin sheath and why does it matter for neuropathy?
The myelin sheath is the fatty insulating coating around nerve fibers that dramatically speeds up signal transmission and supports nerve health. In neuropathy, damage to the myelin sheath slows nerve conduction and causes symptoms like weakness, numbness, tingling, and pain. Some types of neuropathy specifically target myelin (demyelinating neuropathy), while others primarily damage the axon itself.
What is the difference between demyelinating and axonal neuropathy?
Demyelinating neuropathy primarily damages the myelin sheath, slowing nerve conduction while the axon initially remains relatively intact. Axonal neuropathy primarily damages the axon itself, reducing the number of functioning nerve fibers. The distinction matters for treatment: immune-mediated demyelinating neuropathy often responds well to immunotherapy, while axonal damage recovers more slowly and may not respond to immunotherapy.
Can damaged myelin repair itself?
Yes, in the peripheral nervous system, Schwann cells retain the ability to regenerate myelin sheaths throughout life. If the underlying cause of demyelination is stopped or removed — immune attack suppressed, toxin eliminated, deficiency corrected — remyelination can occur and function can recover. However, the regenerated myelin is typically thinner than original, and if the axon itself has been damaged, recovery takes longer and may be incomplete.
How do doctors tell if neuropathy is demyelinating?
Nerve conduction studies are the primary diagnostic tool. Demyelinating neuropathy produces slowed conduction velocity and prolonged latencies, while the amplitude of the signal remains relatively preserved. Axonal neuropathy shows reduced amplitude but relatively normal conduction velocity. Clinical signs like absent deep tendon reflexes and enlarged nerves also suggest demyelination.
Is CIDP a demyelinating neuropathy?
Yes, CIDP is a chronic inflammatory demyelinating polyneuropathy — the condition is literally named for its demyelinating nature. The immune system repeatedly attacks peripheral nerve myelin, causing relapsing or progressive symptoms. Treatment with IVIG, corticosteroids, or plasma exchange suppresses this immune attack and allows remyelination and recovery.
Does myelin damage cause pain?
Yes, myelin damage can cause pain, though it does so through different mechanisms than axonal damage. Partially demyelinated nerve fibers can fire spontaneously, generating false pain signals. Demyelination also makes nerves hypersensitive to normal stimuli. Additionally, as demyelination progresses and eventually causes axonal damage, pain may increase further.
Is multiple sclerosis the same as demyelinating peripheral neuropathy?
No. Multiple sclerosis is a central nervous system demyelinating disease that attacks myelin in the brain and spinal cord. Peripheral demyelinating neuropathy attacks myelin in the peripheral nerves outside the brain and spinal cord. They have different causes, different clinical presentations, different diagnostic tests, and different treatments.
What supplements support myelin health?
B12 is the most critical — deficiency directly causes myelin breakdown and peripheral neuropathy. B1 (thiamine) and B6 at appropriate doses support nerve function generally. Omega-3 fatty acids contribute to myelin membrane structure. Alpha-lipoic acid provides antioxidant protection for nerve tissue. None of these supplements repair severely damaged myelin, but they support the conditions needed for normal myelin maintenance and whatever regeneration is possible.