The first time someone in my support group mentioned cryotherapy, I assumed she meant ice packs. She did not. She meant a $65 session in a chamber that gets to negative two hundred degrees Fahrenheit, where you stand in a swimsuit and wool socks for three minutes while a fog of liquid nitrogen vapor swirls around you. She swore it helped her feet. I went home and started reading.
What I found was both more and less than the marketing brochures promised. Cryotherapy in some forms has real, peer-reviewed evidence for some kinds of neuropathy. In other forms, the evidence is thin, mixed, or actively conflicting. And for people with sensory neuropathy specifically, there are some safety considerations that the wellness-spa industry doesn't always mention.
This is what I learned, and what I'd want a friend with neuropathy to know before they spent the money or stepped into a chamber.
What “Cryotherapy” Actually Means
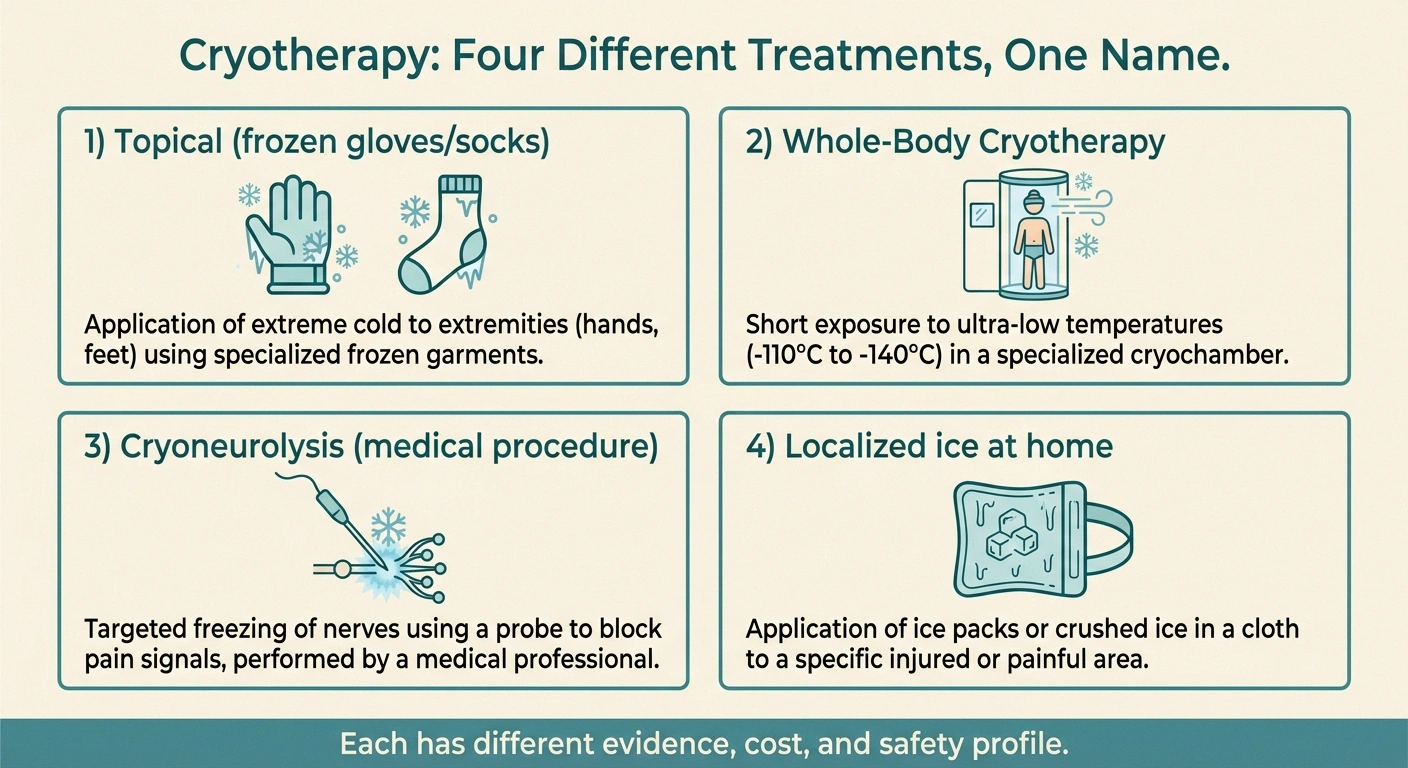

The word covers four very different things, and the evidence base is different for each one. If you don't separate them, you'll either dismiss something that might help or get excited about something that probably won't.
Four Kinds of Cryotherapy — Different Evidence, Different Costs
Topical (frozen gloves/socks)
Strongest evidence base. CIPN prevention during chemo. Often included with infusion.
Cryoneurolysis (medical procedure)
Targeted nerve freezing by a pain physician. Best for specific, identifiable nerve pain. Often insurance-covered.
Whole-Body Cryotherapy (chamber)
Weak evidence in chronic neuropathy. $40-90/session. Treat as an experiment with a budget.
Home ice pack (during flares)
Cheap, reliable, useful for flare-up relief. Use a barrier and a timer.
Key Takeaway
“Cryotherapy” is four very different things. Topical cooling during chemo has the strongest evidence (CIPN prevention). Cryoneurolysis is a real procedure for specific identifiable nerves. Whole-body cryotherapy chambers have weak evidence for chronic neuropathy. A simple ice pack at home is the cheapest and most reliable form for flare-up relief.
Topical cryotherapy. Frozen gloves, frozen socks, ice packs, or cooling sleeves applied directly to the skin. Used most often during chemotherapy infusions to try to prevent nerve damage from drugs like paclitaxel.
Whole-body cryotherapy (WBC). The chamber treatment my friend tried. You stand in a chamber chilled with liquid nitrogen vapor for two to four minutes. Promoted for inflammation, pain, recovery, mood, and a long list of other things.
Cryoneurolysis. A medical procedure performed by a pain specialist, in which a needle-like probe is inserted near a specific nerve and freezes it briefly to interrupt pain signals. This is a real interventional procedure, not a spa treatment.
Localized ice for flare-ups. What most of us already do at home — an ice pack on a hot, painful foot during a flare. Cheap, easy, and worth its own discussion.
Each has a different mechanism, a different evidence base, and a different safety profile. I'll go through them one at a time.
The Strongest Evidence: Cryotherapy During Chemotherapy

If you're going through chemo with a drug like paclitaxel, oxaliplatin, or another agent known to cause nerve damage, this is the corner of the cryotherapy literature where the evidence is most encouraging.
~55%
reduction in CIPN incidence
in pooled analysis of cryotherapy during taxane chemotherapy
Research Says
A 2023 systematic review on cryotherapy for paclitaxel-induced peripheral neuropathy reported meaningful reductions in CIPN incidence and severity, with no serious adverse events across the included studies.
A small cryoneurolysis study of 22 patients with refractory peripheral neuropathic pain reported pain scores dropping from 8.3 at baseline to 2.3 at 1 month and remaining around 5 at 12 months — meaningful long-term relief.
Evidence varies by form. Whole-body cryotherapy in chronic neuropathy has not produced comparably consistent results.
The idea is simple: cold causes blood vessels to constrict, which reduces how much of the chemotherapy drug reaches the small nerves in the hands and feet. Less drug exposure to the nerves means less nerve damage. Patients wear frozen gloves and socks during the infusion and for a short time before and after.
A 2023 systematic review on the safety and efficacy of cryotherapy in preventing paclitaxel-induced neuropathy looked at multiple studies and found that cryotherapy was associated with a meaningful reduction in chemotherapy-induced peripheral neuropathy (CIPN), particularly in sensory symptoms. A George Washington University study in breast cancer patients reached a similar conclusion. A separate systematic review reported that none of the included studies showed serious adverse events from this protocol.
The evidence isn't perfect. Some randomized trials show benefit, others show smaller or no effect, and the optimal duration of cooling is still being worked out. But the overall direction of the evidence is positive enough that some major cancer centers, including Mayo Clinic, are now running formal trials of topical cryotherapy in this population. If you're starting chemotherapy with a drug known to cause neuropathy, this is worth a direct conversation with your oncology team — most centers can accommodate it.
Whole-Body Cryotherapy: The Honest Verdict


Now the harder conversation. Whole-body cryotherapy chambers are a fast-growing wellness business, and the marketing claims for neuropathy are often broader than the evidence supports.
Here's what we can say with reasonable confidence:
- WBC produces a short-term anti-inflammatory and pain-modulating effect in many people. People often report feeling better immediately afterward, sometimes for hours or a day.
- That short-term effect has been studied more in athletes and rheumatologic conditions like ankylosing spondylitis than in peripheral neuropathy specifically.
- Studies on WBC for chronic peripheral neuropathy specifically are scarce, small, and inconsistent.
- Sessions typically cost $40 to $90 each, and the effect doesn't usually last long enough that occasional sessions change the underlying condition.
This doesn't mean it can't help. Some people in my support group have found WBC sessions valuable as part of a broader pain-management plan, especially during flare periods. What it means is that the evidence base is too thin to make confident recommendations, and the cost is high enough that you should treat it as an experiment, not a prescription. If a course of sessions doesn't shift your pain or function in a noticeable way over a month, it's probably not the right tool for you.
One caution: WBC is not appropriate for everyone. People with Raynaud's phenomenon, severe cardiovascular disease, untreated high blood pressure, cold-induced asthma, or cold urticaria (cold allergy) are usually advised against it. Anyone with reduced ability to feel temperature — which is most of us with sensory neuropathy — is at increased risk for cold injury and should be especially careful, no matter what the spa says.
Cryoneurolysis: A Real Procedure for the Right Patient
Cryoneurolysis (sometimes called cryoanalgesia) is the most medically serious option in this article. A pain physician identifies a specific peripheral nerve that is generating pain, inserts a probe under image guidance, and freezes the nerve to a precise temperature for a controlled period. The nerve fibers are interrupted, the pain signal is dampened or stopped, and the nerve regrows over weeks to months. The relief can last anywhere from a few weeks to over a year.
One commonly cited cryoneurolysis study looked at 22 patients with refractory peripheral neuropathic pain and reported pain scores dropping from 8.3 at baseline to 2.3 at one month, 3.2 at three months, 4.7 at six months, and 5.1 at twelve months. That's a meaningful reduction sustained over a year — though only in a small group, and only for patients whose pain could be traced to a specific nerve.
The catch is that cryoneurolysis works best when the pain has a clear anatomical “address” — one identifiable nerve generating the symptoms. Diffuse, length-dependent diabetic neuropathy in both feet does not fit that pattern; it's coming from too many nerves at once. But specific entrapment neuropathies, post-surgical nerve pain, painful neuromas, and isolated peripheral nerve pain syndromes can be excellent candidates.
If you suspect your neuropathy is concentrated in one or two specific nerves rather than diffuse — for example, persistent pain after a knee replacement, a painful neuroma after a foot surgery, or a single-nerve entrapment — it's worth asking your neurologist or pain physician whether cryoneurolysis is on the table for you. It's a real tool, and it's still underused in the United States.
The Quiet Win: Localized Ice at Home

While the headlines go to the chamber and the procedure, the most useful form of cold therapy for many of us is the cheapest one: a regular ice pack on a flare-up foot.
The mechanism is simple. Cold reduces inflammation, slows nerve conduction velocity in the area being cooled, and provides a competing sensory input that can dial down pain perception. None of those effects last more than the duration of the application plus a short tail. But during a bad night, a short tail can be the difference between getting some sleep and not.
Some practical guidance for ice at home:
- Always use a barrier. A thin towel between the ice and the skin. Sensory neuropathy means you cannot reliably feel cold injury, and frozen-gel packs straight on numb feet can cause burns that you only notice after the damage is done.
- Limit each application to fifteen to twenty minutes. Then off for at least an hour before reapplying. Longer is not better — at some point you're just damaging the skin you can't feel.
- Soft gel packs over rigid ones. They contour to the foot and distribute pressure better.
- Watch for color and texture. Healthy skin should pink up within minutes of removing the ice. White, waxy, or hard skin after an application means you went too far.
- Pair with elevation. Cold plus elevation does more than either alone, especially for the swelling and burning components of a flare.
People sometimes ask whether to use heat or cold during a flare. The honest answer is that it varies. Heat works better for some people and some flare types, cold works better for others, and a few people benefit from alternating. Try both in different flares and pay attention to what your body says. Foot soaks of various temperatures can fit into the same general category — experimentation is fair, and you're allowed to figure out what works for your body specifically.
The Cold Paradox: Why Winter Hurts but Ice Sometimes Helps
If you live with neuropathy in a cold climate, you already know that winter weather often makes symptoms worse. Cold weather and neuropathy is one of our most commonly searched topics on this site. So how does that square with cold therapy supposedly helping?
The answer is in the difference between sustained ambient cold and brief, targeted cold.
Sustained cold from winter weather constricts circulation in the feet and hands for hours at a time, which deprives already-struggling nerves of oxygen and nutrients. The cold also stiffens connective tissue, reduces dexterity, and worsens any vascular insufficiency hiding underneath the neuropathy. There's nothing therapeutic about that — it's just slow injury.
Brief targeted cold, on the other hand, produces a short anti-inflammatory effect and then a rebound increase in circulation as the body rewarms the area. The “burn-then-bounce” pattern of a 15-minute ice application is biologically very different from being stuck in a 20-degree parking lot for three hours.
The practical upshot: keeping your feet and hands warm in cold weather is good for your nerves. Brief intentional cold applications during flares is a separate tool, and the two pieces of advice don't contradict each other.
Safety Issues Most People Don't Hear


This section deserves its own headline, because the wellness industry doesn't always lead with it.
Sensory Neuropathy + Cold = Hidden Risk
If you can't reliably feel cold, you can't reliably feel a cold injury. Frostnip and cold burns can develop with applications that wouldn't injure someone with normal sensation.
- Always insulate — thin towel between cold source and skin.
- Set a timer — 15-20 minutes maximum per application.
- Inspect the skin afterward — white, gray, hard, or blistered = cold injury.
- Avoid WBC if you have severe vascular disease, Raynaud's, or cold urticaria.
- Don't fall asleep with an ice pack on. The most common home injury.
Sensory neuropathy compromises your ability to feel cold injury. That sounds obvious until you realize the implication: people with neuropathy can develop frostnip, frostbite, and cold burns from applications that wouldn't injure someone with normal sensation. The skin is doing the same things — turning white, going numb, getting damaged — but the alarm system that normally tells you to stop isn't working.
Some specific cautions:
- Never use dry ice or extremely cold gel packs directly on numb skin. Always insulate.
- Set a timer. Don't rely on “I'll know when it's time to take this off.”
- Inspect the skin afterward. Pink, mottled, or red is normal and resolves quickly. White, gray, hard, or blistered is a cold injury — see your doctor.
- Avoid commercial WBC chambers if you have severe vascular disease or Raynaud's. Those conditions plus extreme cold is a known risk for tissue damage.
- Don't fall asleep with an ice pack. The single most common way home users injure themselves.
None of this is a reason to avoid cold therapy entirely. It's a reason to use it deliberately, with clear time limits, and with respect for the fact that your warning system isn't fully online.
How to Decide if Any of This Is Worth Trying
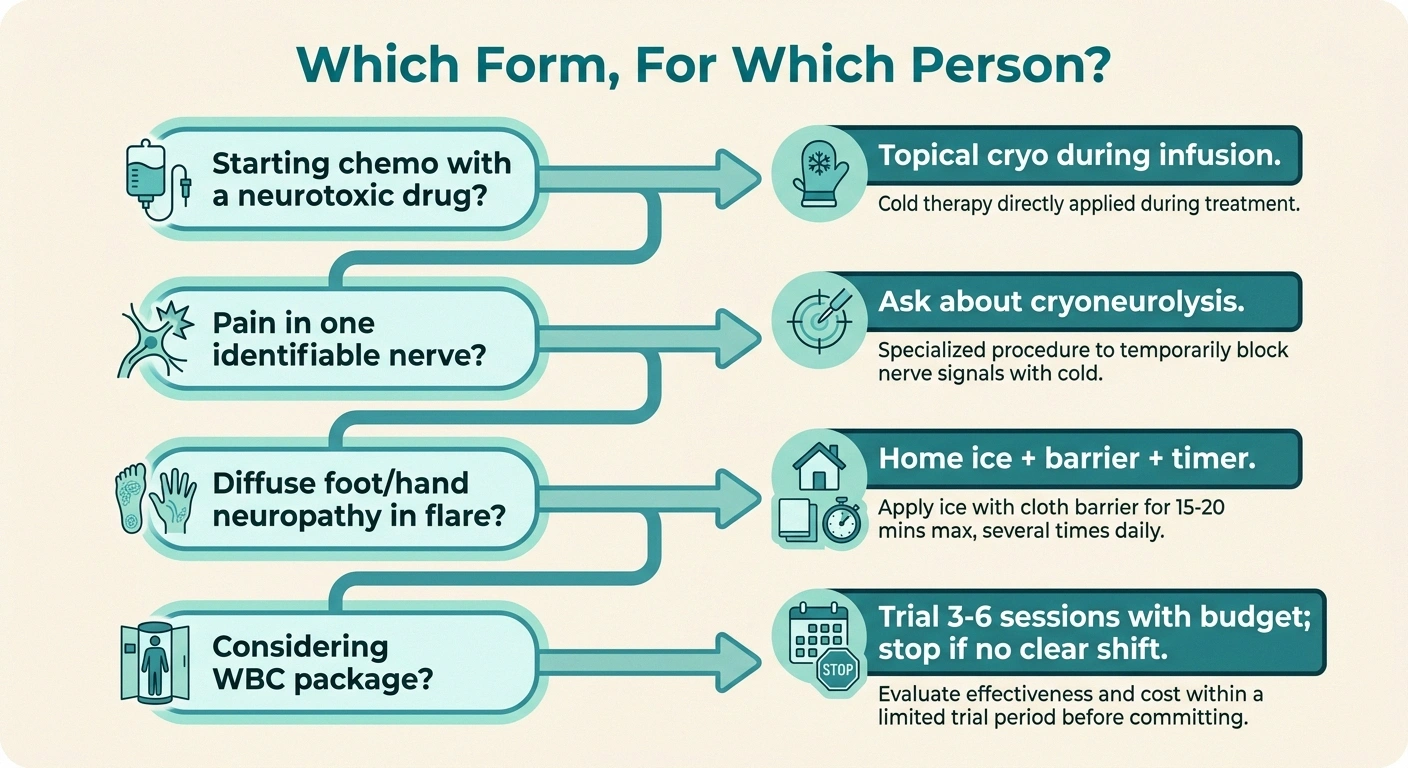
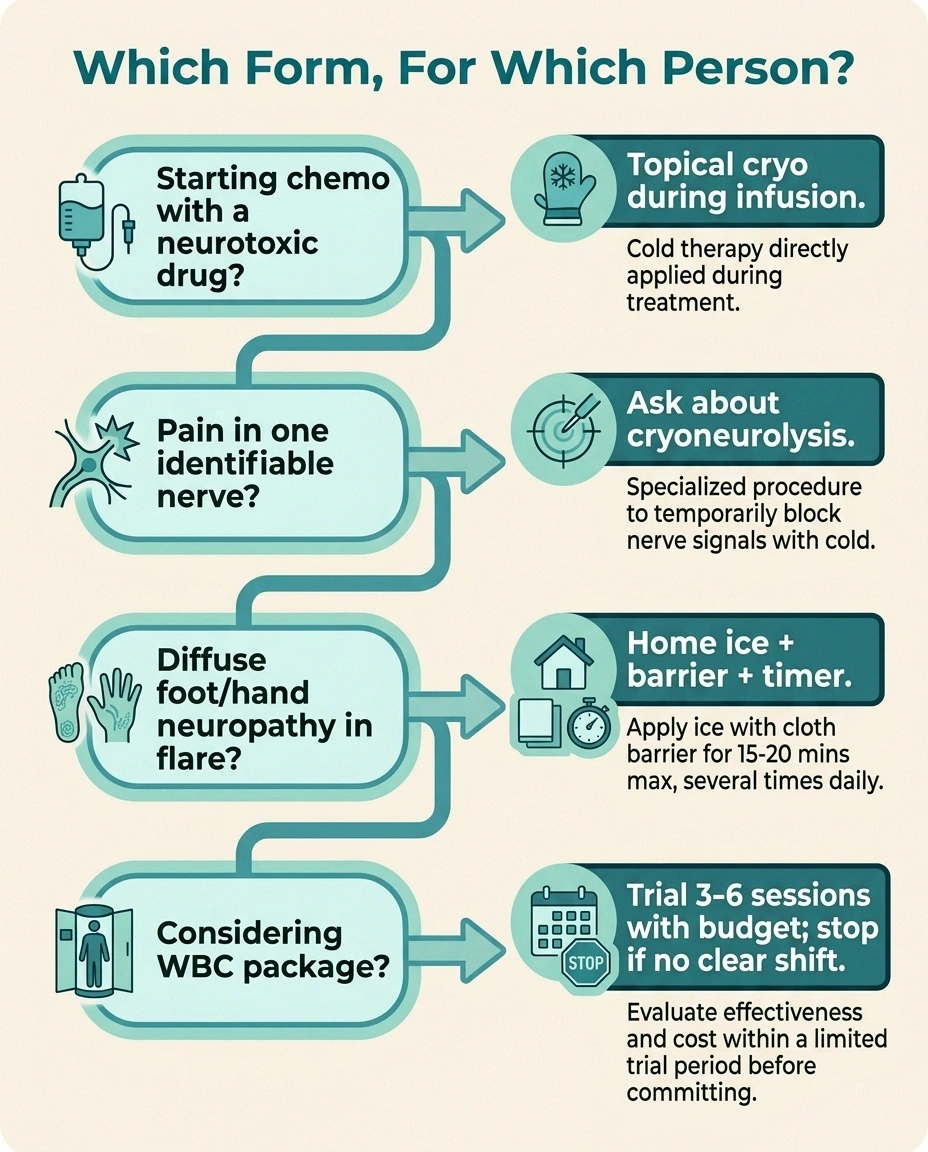
Here's a simple decision framework I use when people in our support group ask me about cryotherapy.
Which Form, For Which Person?
Starting chemo with a neurotoxic drug? Ask oncology about topical cryotherapy gloves/socks during infusion.
Pain in one identifiable nerve? Ask a pain physician about cryoneurolysis.
Diffuse foot/hand neuropathy in flare? Ice pack + barrier + 15-min timer + elevation. Free-ish.
Considering a WBC package? Trial 3-6 sessions with a defined budget. If no clear shift, stop.
If you're starting chemotherapy with a known neurotoxic drug, ask your oncologist about topical cryotherapy gloves and socks during infusion. The evidence is strong enough to be worth trying, and most centers can accommodate it.
If you have flare-up neuropathy and a freezer, a soft gel ice pack with a cloth barrier, applied for 15 minutes during a bad flare, costs essentially nothing and is worth trying. Pair with elevation. Be careful about cold injury.
If you have pain that traces to a specific identifiable nerve — post-surgical, post-traumatic, single-nerve entrapment — ask your pain physician or neurologist about cryoneurolysis. It's a real procedure, it's covered by insurance for the right indication, and it's underused.
If you're considering a whole-body cryotherapy package at a wellness center, treat it as an experiment with a defined budget. Try a course of three to six sessions. If your pain or function does not change in a way you can clearly notice, stop. The evidence base for WBC in chronic peripheral neuropathy is too thin to justify ongoing spending without a clear personal result.
And whatever you try, your overall plan still includes the fundamentals: supporting nerve health from the inside, controlling underlying causes like diabetes, moving daily, sleeping, and managing stress. Cold therapy is a tool. It's not the whole toolbox.
Frequently Asked Questions
Does cryotherapy actually help diabetic neuropathy?
The evidence is weak for whole-body cryotherapy in diabetic peripheral neuropathy specifically. Localized ice for flare-up symptom relief can help in the moment but does not change the underlying disease. The single best evidence for cryotherapy in neuropathy is in preventing chemotherapy-induced nerve damage, not in treating diabetic neuropathy.
Is cryotherapy safe for people with neuropathy?
It can be, with care. The main risk is cold injury, because sensory neuropathy reduces your ability to feel when an application is becoming dangerous. Use barriers between cold sources and skin, set time limits, inspect the skin afterward, and avoid extreme cold if you have severe vascular disease or Raynaud's. People with normal sensation can use cold more aggressively than people with numb feet should.
How is cryotherapy different from just using an ice pack?
An ice pack is technically a form of localized cryotherapy. The major commercial differences are temperature (commercial WBC reaches much colder temperatures briefly) and duration. For most home users with flare-up symptoms, a soft gel pack and a barrier produce most of the practical benefit at a fraction of the cost.
How long does the pain relief from a cryotherapy session last?
For whole-body sessions, typically a few hours to a day for most people who respond. For cryoneurolysis as a medical procedure, the relief can last weeks to over a year, depending on the nerve and the patient. For home ice applications, usually for the duration of the application plus a short tail of an hour or two.
Are there people who should avoid cryotherapy entirely?
Yes. People with Raynaud's phenomenon, severe cardiovascular disease, uncontrolled high blood pressure, cold urticaria (cold allergy), or significant peripheral arterial disease are usually advised against whole-body cryotherapy. People with severe sensory neuropathy should be especially careful with any cold application because of cold-injury risk.
Does insurance cover cryotherapy for neuropathy?
Whole-body cryotherapy at a wellness center is essentially never covered. Cryoneurolysis as a medical procedure performed by a pain physician for an appropriate indication is often covered, but coverage varies and prior authorization is common. Topical cryotherapy during chemotherapy is sometimes provided as part of the infusion at no extra cost.
How does cryotherapy compare to red light or infrared therapy?
The two work by very different mechanisms. Red light therapy aims to stimulate cellular energy production and may support nerve repair. Cryotherapy aims to reduce inflammation and damp pain signaling. Some people use them in different roles — red light as a longer-term repair-supporting tool, cold as a flare-management tool. Neither has the evidence base of pharmacotherapy for established neuropathic pain.
The Bottom Line
Cryotherapy is real medicine in some forms and expensive marketing in others. The strongest case for it is during chemotherapy with a neurotoxic drug, where well-conducted studies show real reductions in nerve damage. The next strongest case is cryoneurolysis for specific, identifiable nerve pain, performed by a qualified pain physician.
For the diffuse, daily, length-dependent neuropathy most people on this site are dealing with, the realistic role of cold therapy is symptomatic. A 15-minute ice pack during a bad flare, used carefully, can take some heat out of the night. A whole-body cryotherapy package may or may not help; treat it like an experiment with a budget and a clear stopping rule.
And whatever you try, give it the same standard you'd give any other treatment: did it actually change your pain or function in a way you can notice and sustain? If yes, keep going. If no, move on. Your time and money belong to the things that actually help.