
Browse all our types and causes of neuropathy guides for more articles like this one.
When most people think about what causes neuropathy, diabetes is usually the first thing that comes to mind. And while diabetes remains the leading cause of peripheral neuropathy, there's a connection that doesn't get nearly enough attention: obesity itself — even without diabetes — may be sufficient to cause nerve damage.
This isn't speculation. A growing body of research, including a major study from the University of Michigan, has found that obese individuals without diabetes have a significantly higher prevalence of neuropathy compared to lean controls — 12.1% versus just 2.2%. And when diabetes is added to the picture, that number jumps to over 40%.
If you're carrying extra weight and experiencing tingling, burning, or numbness in your feet or hands, understanding this connection could be one of the most important things you learn about your condition. Because unlike some causes of neuropathy that can't be reversed, weight is something you can change — and the research suggests that losing weight may actually stabilize nerve damage and improve symptoms.
of obese people without diabetes have neuropathy
of lean controls have neuropathy
of obese people with diabetes have neuropathy
Source: University of Michigan, 2020
The Science: How Excess Weight Damages Nerves
For decades, researchers assumed that when obese people developed neuropathy, it was because of undiagnosed diabetes or prediabetes — elevated blood sugar damaging the nerves over time. But newer research tells a more complex story. Obesity appears to drive nerve damage through multiple independent pathways, some of which have nothing to do with blood sugar levels.
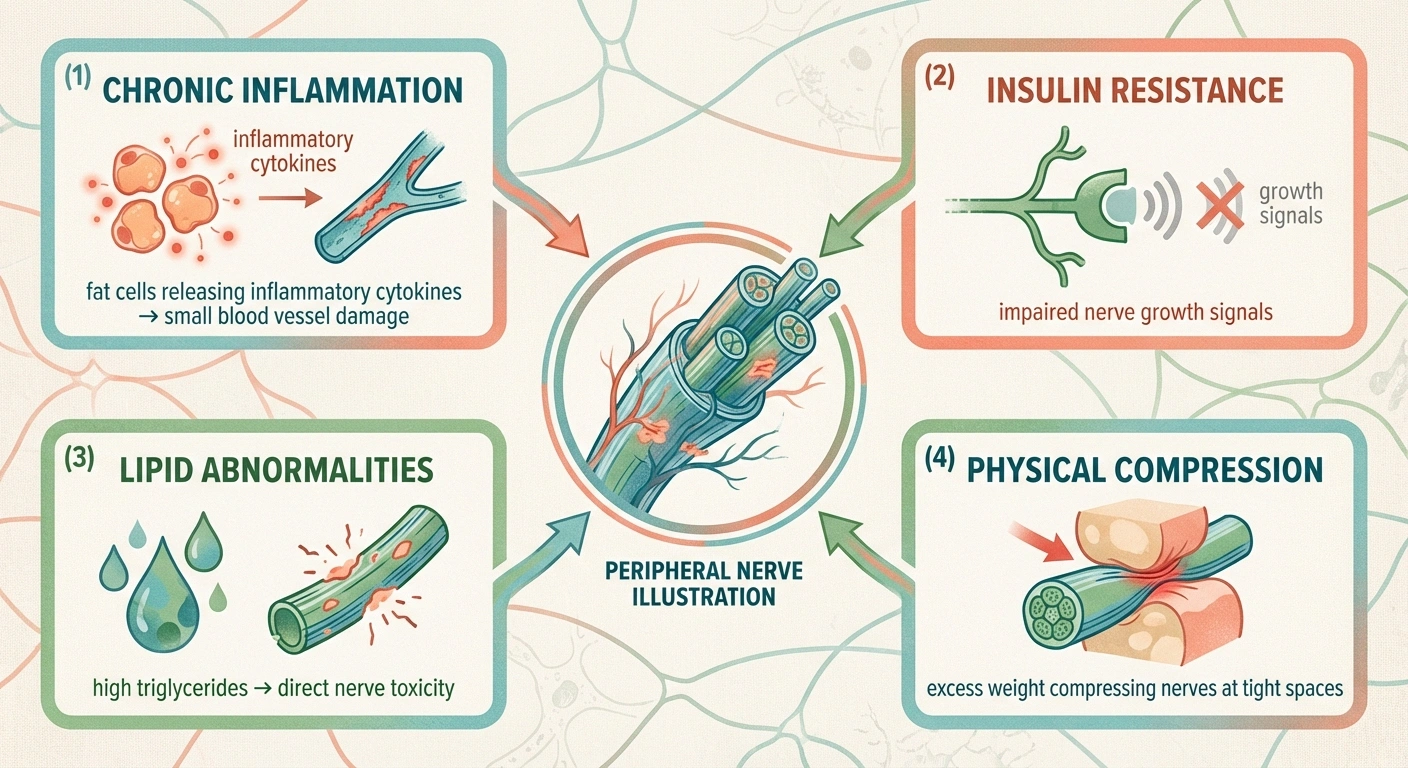
Chronic Low-Grade Inflammation
Excess fat tissue — particularly visceral fat around your midsection — is metabolically active. It constantly releases pro-inflammatory molecules called cytokines and adipokines that circulate throughout your body. Over time, this chronic low-grade inflammation damages the small blood vessels that supply oxygen and nutrients to your peripheral nerves. When nerves don't get adequate blood supply, they begin to deteriorate.
Think of it like a garden hose that's slowly being pinched: the water (oxygen and nutrients) still flows, but not enough to keep everything at the end of the line healthy. Your longest nerves — the ones extending down to your feet — are the most vulnerable because they're the farthest from the heart and rely on the longest, most fragile blood supply chains.
Insulin Resistance (Even Without Diabetes)
Here's something that surprises many people: you don't need to have diabetes — or even prediabetes — for insulin resistance to start affecting your nerves. Obesity commonly causes insulin resistance well before blood sugar levels cross the threshold for a diabetes diagnosis. And there's growing evidence that insulin itself plays a direct role in nerve health, independent of its effect on blood sugar.
Insulin supports nerve growth, repair, and survival. When your cells become resistant to insulin — which happens commonly with obesity — your nerves may not receive the growth signals they need to stay healthy and repair themselves from normal wear and tear. This process is similar to early diabetes-related nerve damage, but it begins before diabetes is ever diagnosed.
Lipid Signaling and Dyslipidemia
High triglycerides and abnormal cholesterol levels — common in obesity — appear to be independent risk factors for neuropathy. A 2022 review published in Antioxidants & Redox Signaling summarized preclinical evidence showing that lipid signaling abnormalities can directly damage peripheral nerves through mechanisms distinct from glucotoxicity (sugar-related damage). Elevated triglycerides were significantly associated with neuropathy in the University of Michigan study, even after controlling for blood sugar levels.
Physical Compression
Excess body weight can physically compress peripheral nerves, particularly in areas where nerves pass through tight spaces. The most well-known example is carpal tunnel syndrome, which is significantly more common in obese individuals. Similarly, excess weight can compress nerves in the lower back (causing sciatica), at the knee (peroneal nerve compression), and at the ankle (tarsal tunnel syndrome).
This type of compression neuropathy is distinct from the metabolic nerve damage described above, but obesity can cause both types simultaneously — creating a double hit on your nerve health.
Central Obesity: It's Not Just About the Number on the Scale
One of the most important findings from recent research is that where you carry your weight matters more than how much you weigh. The University of Michigan study measured nine different body measurements in obese individuals and found something striking: BMI — the number we typically use to define obesity — had no correlation with neuropathy risk. But waist circumference was strongly and significantly associated with neuropathy.

People with larger waist measurements were 39% more likely to have neuropathy for every 10cm increase in waist circumference (OR 1.39, 95% CI 1.10-1.75), even after controlling for other metabolic factors. Meanwhile, people with the same BMI but smaller waists had substantially lower neuropathy rates.
This means that two people who weigh exactly the same could have very different neuropathy risks based on their body fat distribution. Central obesity — fat concentrated around your abdomen — produces more inflammatory cytokines and creates more metabolic disruption than fat distributed elsewhere on the body.
The practical implication: even modest reductions in waist circumference may reduce your neuropathy risk more than overall weight loss measured by BMI alone. This is encouraging because targeted exercises, dietary changes, and lifestyle modifications can reduce central adiposity even when total weight loss is modest.
Research Says
Waist circumference — not BMI — predicts neuropathy risk. Every 10cm increase in waist measurement was associated with a 39% higher chance of neuropathy (OR 1.39, 95% CI 1.10-1.75). Two people at the same weight can have very different neuropathy risks based on where they carry their fat.
The Metabolic Syndrome Connection
Obesity rarely exists in isolation. It's typically accompanied by a cluster of metabolic problems known as metabolic syndrome — the combination of central obesity, high blood pressure, high triglycerides, low HDL cholesterol, and elevated blood sugar. Each component of metabolic syndrome independently increases neuropathy risk, and together they create a synergistic effect.
According to a 2024 review in the Journal of Clinical & Translational Endocrinology, metabolic syndrome drives neuropathy through progressive mitochondrial dysfunction in both neurons and their axons. Mitochondria are the energy factories of your cells, and when they malfunction, nerves — which are among the most energy-demanding cells in your body — suffer disproportionately.
The systolic blood pressure component deserves special attention. In the University of Michigan study, higher systolic blood pressure was the strongest metabolic predictor of neuropathy (OR 2.89, 95% CI 1.49-5.61) — even stronger than waist circumference. High blood pressure damages the small blood vessels feeding your nerves, creating yet another pathway for nerve injury.
This is why addressing neuropathy in the context of obesity isn't just about losing weight. Managing blood pressure, reducing triglycerides, and improving overall metabolic health are all important pieces of protecting your nerves.
Can Weight Loss Improve Neuropathy?
This is the question everyone wants answered — and the research is cautiously encouraging.

A landmark 2021 study published in Obesity followed 131 severely obese participants (with and without diabetes) through a medically supervised weight loss program for two years. Participants lost an average of 12.4 kg (about 27 pounds) through a structured dietary intervention. The results:
- Nerve fiber density stabilized. Intraepidermal nerve fiber density (IENFD) — the most objective measure of small fiber nerve health — remained stable over two years. Given that natural history studies show IENFD typically decreases over time in obese individuals, stabilization is a significant positive finding.
- Symptoms improved. Neuropathy symptom scores on the Michigan Neuropathy Screening Instrument questionnaire improved significantly.
- Quality of life improved. Participants showed improvements in two NeuroQoL (quality of life) subdomains.
- All metabolic parameters improved — triglycerides, blood sugar, cholesterol — except blood pressure.
The lead researcher, Dr. Brian Callaghan of the University of Michigan, summarized: “Dietary weight loss stabilizes or slightly improves neuropathy outcomes in obese patients with or without diabetes.”
It's important to set realistic expectations. Weight loss does not appear to reverse existing nerve damage in the short term — but it may halt progression and improve how you feel. For many people with neuropathy, stopping the decline is a meaningful victory. And some participants did see improvement in objective nerve measures, suggesting that given enough time and sustained weight loss, some degree of nerve recovery may be possible.
Key Takeaway
Weight loss may not reverse existing nerve damage, but it can halt progression and improve symptoms. After 2 years of dietary weight loss, nerve fiber density stabilized and symptom scores improved significantly — a meaningful victory when the natural course is continued decline.
A Word of Caution: Rapid Weight Loss and Nerve Damage
Before you begin an aggressive weight loss program, there's an important caution to be aware of. Rapid weight loss can itself cause a specific type of neuropathy — a condition sometimes called “slimmer's paralysis” or peroneal neuropathy following weight loss.

A 2024 case study published in the Journal of Geriatric Physical Therapy described a 49-year-old woman who developed foot drop after extreme weight loss over 6 months while taking a GLP-1 receptor agonist (like Ozempic or Mounjaro) for obesity management. The rapid loss of protective fat around the knee exposed the peroneal nerve to compression, causing significant nerve damage.
This doesn't mean weight loss is dangerous — it means the pace of weight loss matters. Gradual, sustained weight loss (1-2 pounds per week) gives your body time to adapt and is far safer for your nerves. This is especially important if you're already dealing with neuropathy, as your nerves are more vulnerable to additional insults.
If you're considering bariatric surgery or using weight loss medications, discuss the potential impact on your nerves with both your prescribing doctor and your neurologist.
Talk to Your Doctor
If you're considering bariatric surgery, GLP-1 medications (Ozempic, Wegovy, Mounjaro), or very-low-calorie diets, discuss the pace of weight loss with your neurologist. Gradual weight loss of 1-2 pounds per week is significantly safer for your nerves than rapid loss.
Practical Steps for Managing Weight With Neuropathy
Losing weight when you have neuropathy comes with unique challenges. Pain in your feet can make exercise difficult. Medications like gabapentin and pregabalin can cause weight gain as a side effect. And the emotional toll of living with chronic pain can drive stress eating. Here's how to work with these challenges rather than against them:
Exercise Options That Work With Neuropathy
The research consistently shows that both diet and exercise contribute to neuropathy improvement, but you need to choose activities that don't worsen your symptoms:

- Swimming and water exercises: Water supports your body asking about medication weight gain risks, eliminates impact on painful feet, and provides resistance for muscle building. This is often the best exercise option for people with significant foot neuropathy.
- Recumbent cycling: Takes pressure off your feet while providing cardiovascular exercise and lower body movement.
- Chair-based exercises: Seated strength training allows you to work your muscles without putting stress on numb or painful feet.
- Gentle yoga: Improves balance, flexibility, and strength while also addressing the mental health aspects of living with chronic pain.
- Walking (if tolerable): Even short walks with proper supportive shoes can make a difference. Start with what you can manage and gradually increase.
The University of Michigan weight loss study included a recommendation of 40 minutes daily of moderate activity during the initial intensive phase, increasing to 60 minutes during maintenance. But any increase from your current activity level is beneficial — even 10 minutes of movement is better than none.
Dietary Approaches
For neuropathy specifically, diet may be even more important than exercise for weight management. The University of Michigan study achieved significant results primarily through dietary intervention (structured meal replacement at 800 kcal/day initially, transitioning to 1,200-1,500 kcal/day). While you don't need to follow that exact protocol, the principle is clear: dietary changes drive the bulk of weight loss.
A neuropathy-friendly diet should focus on:
- Anti-inflammatory foods: Fruits, vegetables, fatty fish, nuts, and olive oil reduce the chronic inflammation that drives obesity-related nerve damage.
- Blood sugar stability: Even without diabetes, choosing foods that don't spike blood sugar (whole grains over refined, fiber with every meal) protects your nerves.
- B-vitamins and key nutrients: Ensure adequate intake of B12, folate, and other vitamins critical for nerve health. Calorie restriction can inadvertently cause nutrient deficiencies that worsen neuropathy.
- Reduced processed foods and added sugars: These drive both inflammation and metabolic dysfunction.
Addressing Medication-Related Weight Gain

If you're taking gabapentin, pregabalin, or certain antidepressants for neuropathy pain, weight gain is a common side effect that can create a frustrating cycle — the medication that treats your nerve pain also contributes to the weight that damages your nerves.
Talk to your doctor about this explicitly. Options may include:
- Switching to a neuropathy medication with a more neutral weight profile (like duloxetine, which is the relationship between weight changes and neuropathy-neutral or may promote modest weight loss)
- Adjusting your dose to find the minimum effective amount
- Adding non-medication pain management strategies (like TENS therapy or physical therapy) to reduce reliance on weight-promoting medications
The Gut Microbiome: An Emerging Connection

One of the most fascinating areas of emerging research is the connection between the gut microbiome, obesity, and neuropathy. Preclinical studies summarized in the 2022 Antioxidants & Redox Signaling review have found that fecal transplants from lean animals to obese animals can improve neuropathy outcomes. Butyrate — a short-chain fatty acid produced by healthy gut bacteria — appears to have direct neuroprotective effects.
While this research is still in its early stages and no clinical treatments have emerged yet, it suggests that the health of your gut may influence the health of your nerves through mechanisms we're only beginning to understand. Eating a fiber-rich diet that supports a diverse gut microbiome may offer nerve-protective benefits beyond its effects on weight and inflammation.
Frequently Asked Questions

Can obesity cause neuropathy without diabetes?
Yes, research from the University of Michigan found that 12.1% of obese individuals with completely normal blood sugar had neuropathy, compared to just 2.2% of lean controls. Obesity appears to damage nerves through chronic inflammation, insulin resistance (which exists before diabetes develops), lipid abnormalities, and physical compression — mechanisms independent of high blood sugar.
How much weight do I need to lose to help my neuropathy?
The University of Michigan study showed neuropathy stabilization and symptom improvement with an average weight loss of about 27 pounds over two years. However, even smaller amounts of weight loss can improve the metabolic factors that drive nerve damage. Reducing waist circumference may be more important than total pounds lost, so focus on lifestyle changes that reduce central obesity specifically.
Will losing weight reverse neuropathy damage?
Current research shows that weight loss can stabilize nerve fiber density (halt further damage) and improve neuropathy symptoms and quality of life. Full reversal of existing nerve damage has not been consistently demonstrated in studies, but halting progression is clinically meaningful. Some participants in weight loss studies did show improvement in objective nerve measures, suggesting partial recovery may be possible with sustained weight loss over time.
Can rapid weight loss cause neuropathy?
Yes, very rapid weight loss can cause a condition called peroneal neuropathy or slimmer's paralysis, where rapid loss of protective fat around the knee exposes the peroneal nerve to compression. This has been documented with extreme dieting, bariatric surgery, and more recently with GLP-1 receptor agonist medications. Gradual weight loss of 1-2 pounds per week is safer for your nerves.
Does BMI or waist size matter more for neuropathy risk?
Waist circumference is a much stronger predictor of neuropathy risk than BMI. In the University of Michigan study, BMI showed no significant difference between obese people with and without neuropathy, but waist circumference was significantly associated with neuropathy risk. Central obesity — fat around the abdomen — drives more inflammation and metabolic disruption than fat distributed elsewhere on the body.
Do weight loss medications affect neuropathy?
This is an active area of research. GLP-1 receptor agonists like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro) can promote significant weight loss, which may benefit neuropathy through improved metabolic health. However, the rapid weight loss these medications can cause has been associated with compression neuropathy in case reports. If you're considering these medications and have neuropathy, discuss the pace of weight loss with your neurologist.
The Bottom Line
The relationship between weight and nerve health is more direct and more important than most people realize. Obesity damages nerves through chronic inflammation, insulin resistance, lipid abnormalities, and physical compression — and these effects can begin long before diabetes is ever diagnosed. But the encouraging counterpoint is that weight loss, even modest amounts, may stabilize nerve damage and improve the symptoms that affect your daily life.
If you're dealing with both obesity and neuropathy, I'd encourage you to have an honest conversation with your doctor about how your weight may be contributing to your nerve symptoms. Focus on gradual, sustainable changes — not crash diets — and pay particular attention to reducing central adiposity (waist circumference). The combination of an anti-inflammatory diet, neuropathy-friendly exercise, and medical management of metabolic risk factors gives you the best chance of protecting the nerves you have and potentially improving how you feel.
This article is for informational and educational purposes only. Always consult with your healthcare provider before making changes to your diet, exercise routine, or medication regimen. Weight loss should be supervised by a healthcare professional, especially if you have neuropathy or other medical conditions.

