
Neuropathy After COVID: What Long Haulers Need to Know
When the messages started coming in during 2021 and 2022, I didn't know what to make of them. Readers in their 30s and 40s — people with no prior history of nerve problems — describing tingling in their feet, burning sensations in their hands, and numbness that appeared weeks or months after recovering from COVID-19. “I had a mild case,” one woman named Sarah wrote. “I was sick for three days. Now, eight months later, I can barely feel my toes.”
Since then, the research has caught up with what these readers were experiencing. COVID-19 can cause peripheral neuropathy — and it can do so even after mild infections, even in people with no pre-existing risk factors. A large-scale study published in Nature Medicine found that former COVID patients had a 42% increased risk of neurological problems one year after infection compared to uninfected controls. Small fiber neuropathy, in particular, has emerged as one of the more common neurological complications of long COVID.
If you're dealing with nerve symptoms that appeared after a COVID infection, this article covers what the research currently shows, what types of neuropathy are linked to COVID, what testing to ask for, and what treatments may help. This field is still evolving — but there's enough solid evidence now to guide you toward answers.
How COVID-19 Damages Peripheral Nerves
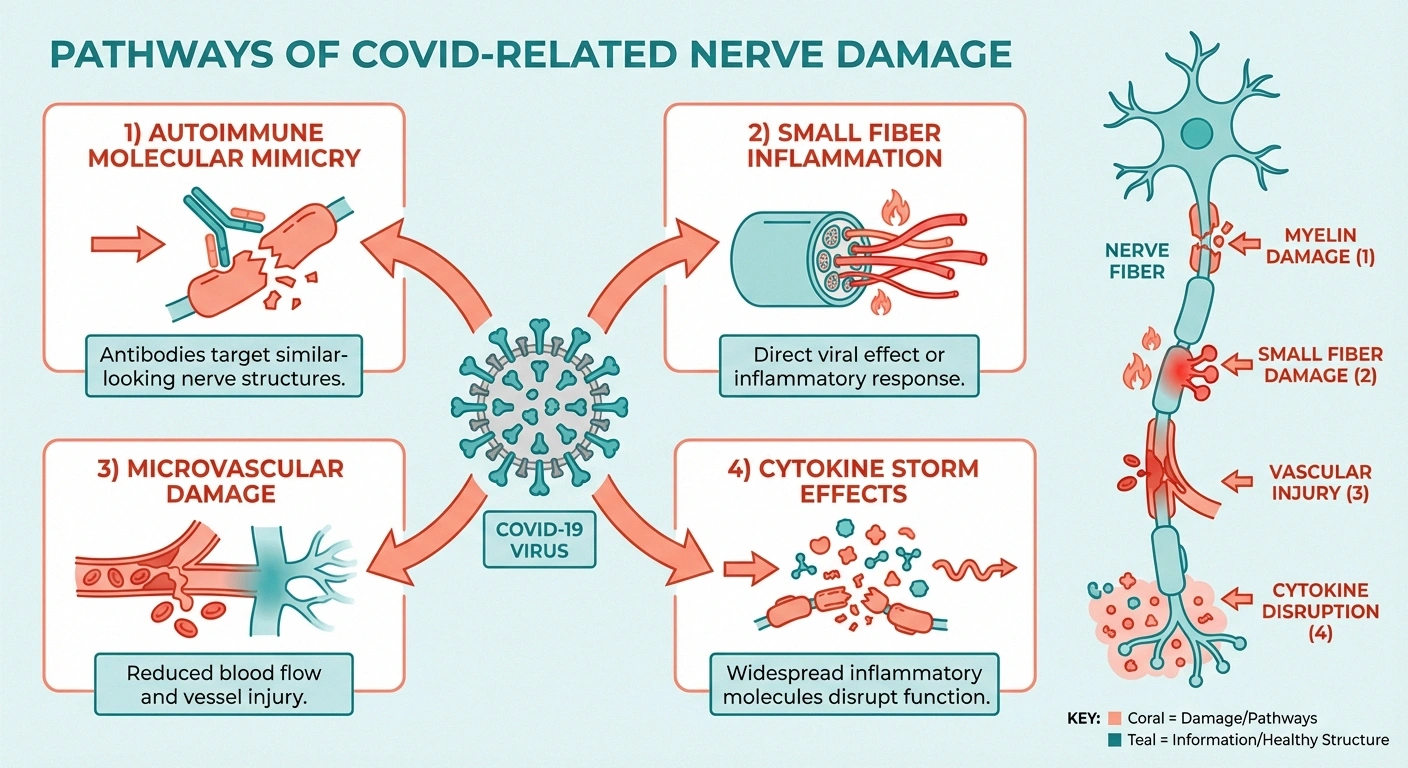
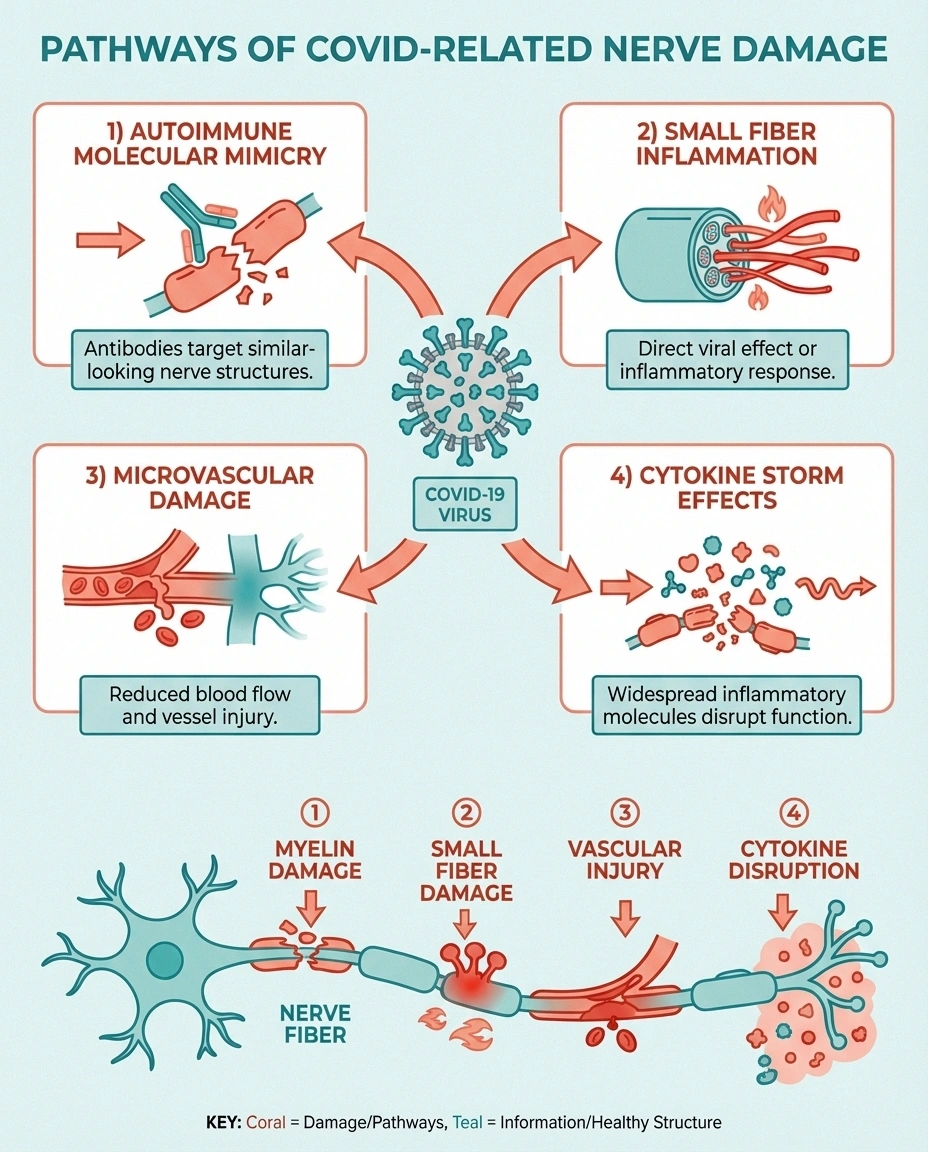
Researchers have identified several mechanisms by which SARS-CoV-2 can damage the peripheral nervous system. Unlike many traditional causes of neuropathy, COVID-related nerve damage often involves the immune system attacking nerves rather than the virus infecting them directly.
Increased neurological risk 1 year after COVID (Nature Medicine)
Of long COVID neuro patients had small fiber neuropathy on biopsy (Harvard/MGH)
Post-infectious autoimmune response. This is the most widely supported mechanism. The virus triggers an immune response that, in some people, begins attacking the body's own nerve tissue — a process called molecular mimicry. Components of the SARS-CoV-2 spike protein resemble proteins found on peripheral nerves, confusing the immune system into targeting them. This is similar to the mechanism behind Guillain-Barré syndrome, which has also been reported after COVID infection.
Small fiber neuropathy from inflammation. A 2022 study from Massachusetts General Hospital and Harvard Medical School found that 59% of long COVID patients with neurological symptoms had evidence of small fiber neuropathy on skin biopsy. The researchers found reduced intraepidermal nerve fiber density — the hallmark of small fiber damage — even in patients whose standard nerve conduction studies were normal. The study attributed this to immune-mediated nerve damage rather than direct viral invasion.
Microvascular damage. COVID-19 causes widespread damage to small blood vessels throughout the body — including the vasa nervorum that feed peripheral nerves. This endotheliopathy (damage to the cells lining blood vessels) reduces oxygen and nutrient delivery to nerve fibers, leading to ischemic nerve damage similar to what occurs in diabetic neuropathy.
Direct viral neurotropism. While less common than immune-mediated damage, there is evidence that SARS-CoV-2 can directly infect nerve cells through the ACE2 receptor, which is expressed on some peripheral nerve tissues. However, most researchers believe this is a minor pathway compared to the autoimmune and vascular mechanisms.
Cytokine storm effects. Severe COVID infections produce a massive release of inflammatory cytokines — including TNF-α, IL-6, and IL-1β — that can damage peripheral nerves directly. Even after the acute infection resolves, some patients continue to have elevated cytokine levels for months, perpetuating nerve damage.
Types of Neuropathy Linked to COVID
Several distinct patterns of nerve damage have been documented after COVID infection:
Emergency Warning
Guillain-Barré syndrome (GBS) can develop within weeks of COVID infection and causes rapidly progressing weakness starting in the legs. If you experience rapid-onset weakness moving upward through your body after COVID, seek emergency medical attention immediately. GBS is treatable but requires urgent care.
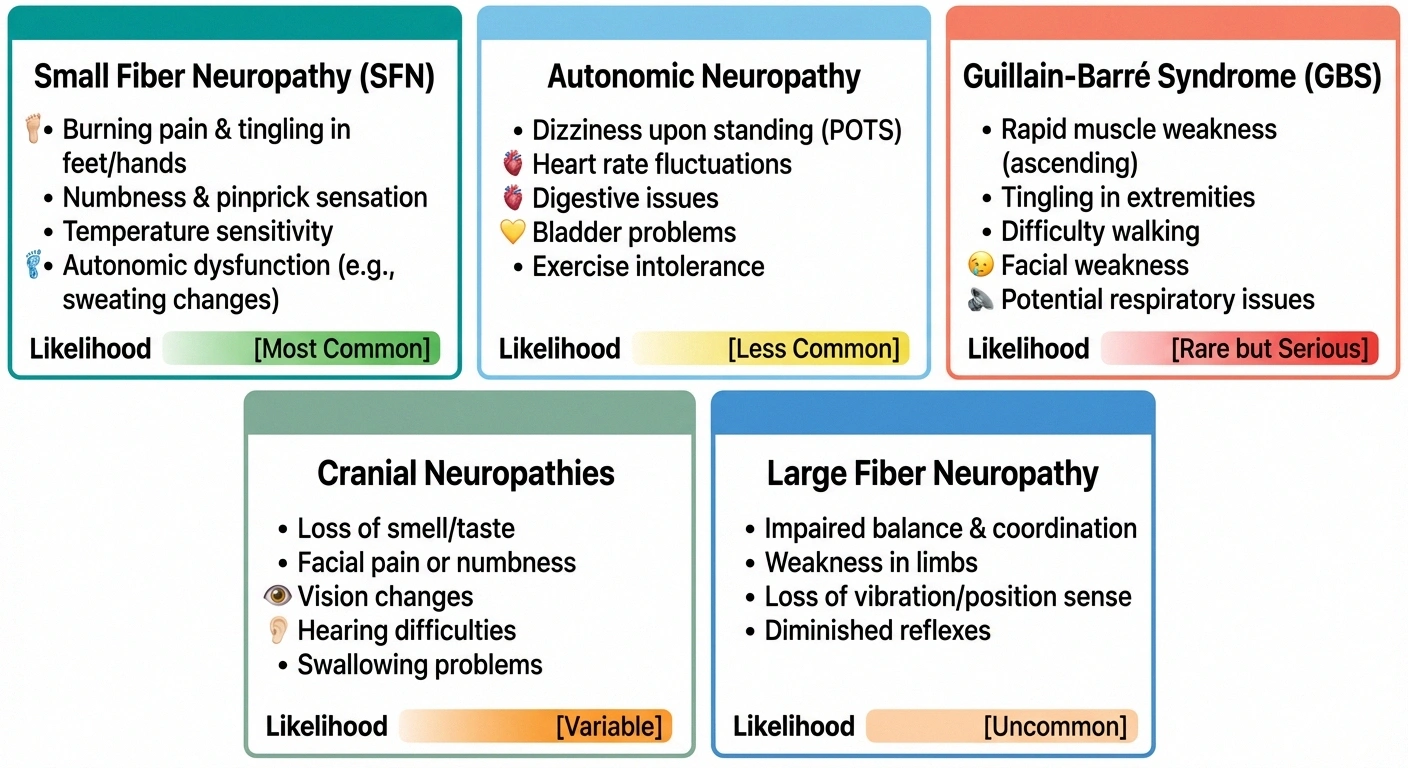
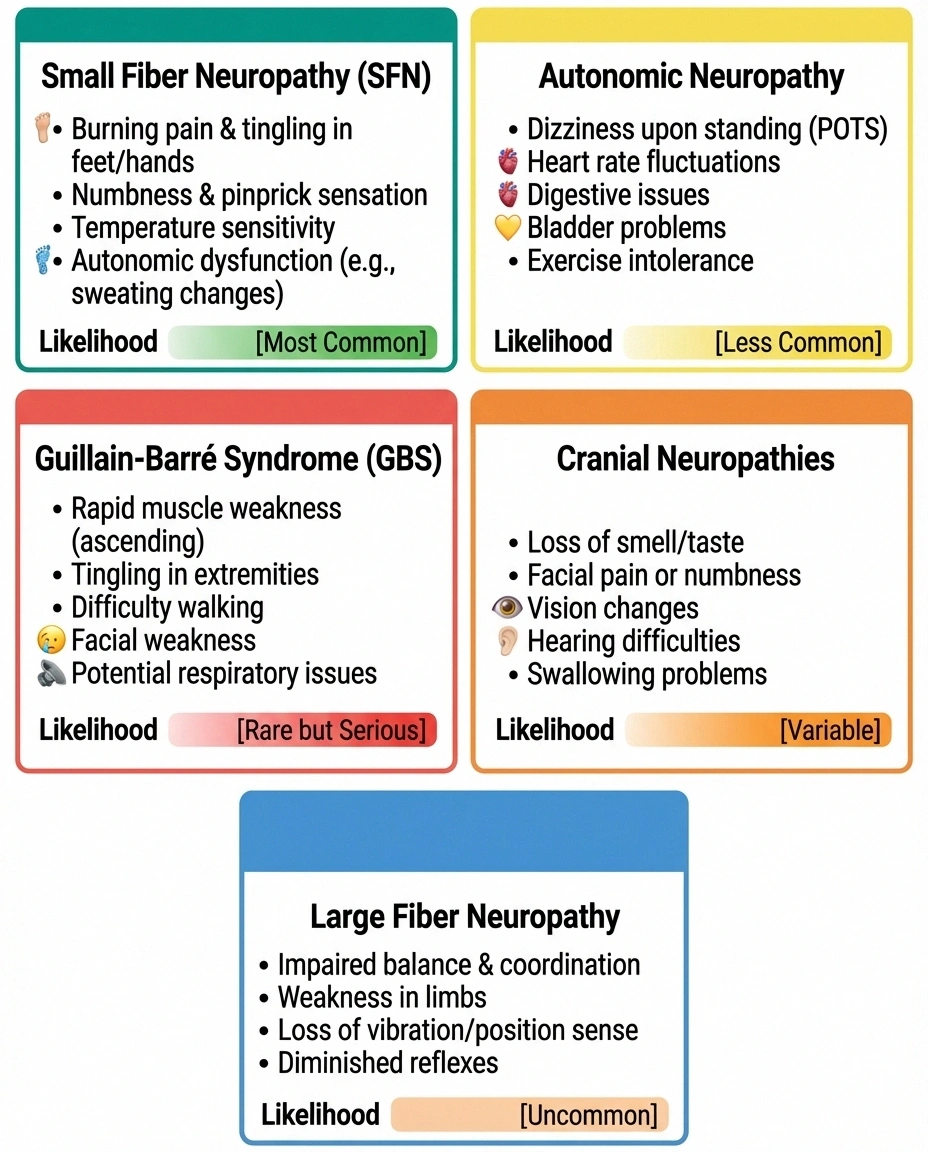
Small fiber neuropathy (most common). Affects the thin, unmyelinated nerve fibers that sense pain, temperature, and carry autonomic signals. Symptoms include burning pain, tingling, hypersensitivity to touch, and temperature dysregulation. Standard EMG/nerve conduction studies are often NORMAL because they only test large fibers. A skin punch biopsy is needed for diagnosis.
Autonomic neuropathy. Damage to the nerves controlling involuntary functions: heart rate, blood pressure, digestion, sweating, and bladder function. Many long COVID symptoms — including POTS (postural orthostatic tachycardia syndrome), dizziness upon standing, abnormal sweating, and digestive problems — may actually be autonomic neuropathy caused by COVID-related nerve damage.
Guillain-Barré syndrome (GBS). An acute autoimmune attack on peripheral nerves causing rapid-onset weakness, typically starting in the legs and moving upward. GBS cases have been reported within weeks of COVID infection. While rare, it requires emergency medical treatment. If you experience rapidly progressing weakness after COVID, seek immediate medical attention.
Cranial neuropathies. Damage to the nerves of the face and head. Loss of smell and taste (anosmia and ageusia) are the most recognized cranial nerve symptoms of COVID, but facial numbness, visual changes, and hearing problems have also been reported.
Large fiber neuropathy. Less common than small fiber involvement, but some patients develop numbness, weakness, and reduced reflexes consistent with large fiber damage detectable on standard nerve conduction studies.
Who Is at Risk?
Post-COVID neuropathy doesn't only affect people who were hospitalized or severely ill. Research consistently shows that nerve complications can develop after mild COVID infections. However, certain factors appear to increase risk:
- Severity of acute infection: More severe COVID illness correlates with higher neurological complication rates, but mild cases can also trigger neuropathy
- Pre-existing autoimmune conditions: People with existing autoimmune tendencies may be more susceptible to post-infectious immune-mediated nerve damage
- Pre-existing diabetes or metabolic syndrome: These conditions already stress peripheral nerves, making them more vulnerable to additional COVID-related damage
- Timing: Neurological symptoms most commonly appear 2-12 weeks after infection, consistent with a delayed autoimmune mechanism
- Reinfection: Some patients report worsening or new neurological symptoms after repeat COVID infections
Getting Diagnosed: What Tests to Ask For
One of the biggest challenges with post-COVID neuropathy is that standard neurological tests may come back normal — particularly for small fiber neuropathy, which is the most common type. Here's what to discuss with your neurologist:
Critical Testing Tip
Standard EMG and nerve conduction studies often come back normal in post-COVID neuropathy because they only test large fibers. The most common type (small fiber neuropathy) requires a skin punch biopsy. If your basic tests are normal but symptoms persist, specifically request this test.
- Skin punch biopsy: The gold standard for diagnosing small fiber neuropathy. A small skin sample (usually from the ankle and thigh) is examined for intraepidermal nerve fiber density. This is the test that the Harvard/MGH study used to identify SFN in long COVID patients.
- Nerve conduction study (NCS) and EMG: Tests large fiber function. May be normal in small fiber neuropathy but should still be performed to rule out large fiber involvement or other conditions.
- Autonomic function testing: Quantitative sudomotor axon reflex test (QSART), tilt table testing, and heart rate variability analysis can identify autonomic neuropathy.
- Blood work: Comprehensive panel including B12, folate, thyroid function, fasting glucose, HbA1c, ANA (antinuclear antibodies), and ESR/CRP (inflammation markers). This rules out other treatable causes.
- Ganglioside antibodies: Specific antibodies associated with autoimmune neuropathies (anti-GM1, anti-GQ1b, etc.) may be positive in post-COVID autoimmune neuropathy.
If your doctor dismisses your symptoms because basic blood work and EMG are normal, specifically request a skin punch biopsy. The Harvard study found that this was the test most likely to reveal COVID-related nerve damage. For general guidance on navigating this process, our guide on talking to your doctor about neuropathy may help.
Treatment Options
Treatment for post-COVID neuropathy depends on the underlying mechanism, but several approaches have shown promise:
Recovery Timeline
Many patients with post-COVID neuropathy experience gradual improvement over 6-18 months as the immune response normalizes. Small nerve fibers regenerate at roughly 1 inch per month. This isn't guaranteed, but the trajectory for many people is slow improvement rather than permanent disability.
Immunotherapy (for immune-mediated cases). When neuropathy is driven by an autoimmune response, treatments that modulate the immune system can be effective. Intravenous immunoglobulin (IVIG) has been used successfully in some post-COVID neuropathy patients, particularly those with Guillain-Barré syndrome or documented autoimmune markers. Corticosteroids may also be used for short-term immune suppression. These treatments require specialist oversight.
Pain management. The same medications used for other forms of neuropathic pain can help with post-COVID neuropathy: gabapentin, pregabalin, duloxetine, and amitriptyline. Low-dose naltrexone (LDN) has also been explored in some long COVID treatment protocols for its anti-inflammatory and pain-modulating properties.
Anti-inflammatory approaches. Because neuroinflammation plays a central role, an anti-inflammatory diet, alpha-lipoic acid supplementation, and regular exercise may help reduce the ongoing inflammatory burden on nerves. These are supportive rather than curative, but addressing chronic inflammation is always beneficial for nerve health.
Physical therapy and rehabilitation. Physical therapy can help maintain function, improve balance, and reduce pain through targeted exercises. For autonomic symptoms like POTS, graduated exercise programs and increased salt/fluid intake are often first-line treatments.
Time. Many patients with post-COVID neuropathy experience gradual improvement over 6-18 months as the immune response normalizes and nerve fibers slowly regenerate. This isn't guaranteed, but the trajectory for many people is one of slow improvement rather than permanent disability.
What About Vaccination and Neuropathy?
This is a question many readers have asked. Post-vaccination neuropathy has been reported in the medical literature, though it appears to be rare. The mechanism is believed to be similar to post-infection neuropathy — an autoimmune response triggered by the spike protein produced by the vaccine. Reports to VAERS (the Vaccine Adverse Event Reporting System) and peer-reviewed case series have documented small fiber neuropathy and Guillain-Barré syndrome following COVID vaccination.
However, the risk of neurological complications from COVID infection itself is significantly higher than from vaccination. The Nature Medicine study found a 42% increased neurological risk from infection, while vaccine-associated neurological events are estimated at orders of magnitude lower. The decision about vaccination is personal and should be discussed with your neurologist, especially if you have a history of autoimmune neuropathy or Guillain-Barré syndrome.
Living With Post-COVID Neuropathy
The day-to-day management of post-COVID neuropathy is similar to managing neuropathy from other causes, with a few unique considerations:

Your Post-COVID Neuropathy Action Plan
Get a neurologist referral (ideally neuroimmunology or long COVID specialist)
Request comprehensive blood work (B12, thyroid, HbA1c, ANA, CRP/ESR)
Ask for skin punch biopsy if EMG is normal but symptoms persist
Start a symptom journal tracking pain, numbness, and triggers
Discuss treatment options (pain management, immunotherapy if indicated)
Connect with a long COVID or neuropathy support community
- Pace yourself. Post-exertional malaise — symptom worsening after physical or mental activity — is common in long COVID. If your neuropathy symptoms flare after activity, you may need to carefully titrate your exercise and daily activities to avoid triggering crashes.
- Monitor for improvement. Keep a symptom journal tracking pain levels, numbness, and functional ability. Many people see gradual improvement that's hard to notice day-to-day but becomes apparent over months.
- Address the mental health component. Anxiety and depression are extremely common in long COVID, and they can amplify pain perception. Treating the emotional side isn't separate from treating the neuropathy — it's part of the same recovery.
- Connect with others. Support groups — both neuropathy-specific and long COVID communities — provide validation and practical advice. You're not imagining your symptoms, and you're not alone.
- Stay informed but discerning. The research on long COVID and neuropathy is evolving rapidly. Stick to peer-reviewed sources and discussions with your medical team rather than social media anecdotes.
For more guidance on day-to-day management, explore our living with neuropathy resources.
Frequently Asked Questions
How common is neuropathy after COVID?
The exact prevalence varies by study, but research from Massachusetts General Hospital found that 59% of long COVID patients with neurological symptoms had evidence of small fiber neuropathy on skin biopsy. Among the broader population of COVID survivors, the Nature Medicine study found a 42% increased risk of neurological problems compared to uninfected controls. Not all of these are neuropathy specifically, but peripheral nerve involvement is one of the more common long COVID neurological complications.
Can post-COVID neuropathy go away on its own?
Many patients experience gradual improvement over 6 to 18 months, though the timeline varies widely. Some people recover fully, particularly those with mild symptoms and no pre-existing risk factors. Others experience persistent symptoms that require ongoing management. The autoimmune mechanism suggests that once the aberrant immune response subsides, nerve regeneration can begin — but small nerve fibers regenerate slowly, at roughly 1 inch per month.

My EMG and nerve conduction tests were normal. Does that mean I don't have neuropathy?
Not necessarily. Standard EMG and nerve conduction studies only test large myelinated nerve fibers. The most common type of post-COVID neuropathy is small fiber neuropathy, which requires a skin punch biopsy for diagnosis. If your standard tests are normal but you have symptoms consistent with neuropathy — burning pain, tingling, temperature sensitivity, autonomic dysfunction — ask your neurologist specifically about a skin biopsy.
Is post-COVID neuropathy the same as diabetic neuropathy?
They share some symptoms but have different underlying causes. Diabetic neuropathy is driven primarily by sustained high blood sugar damaging the vasa nervorum over years. Post-COVID neuropathy is typically driven by a post-infectious autoimmune response and inflammation that develops over weeks to months. The treatment approach differs — diabetic neuropathy management centers on blood sugar control, while post-COVID neuropathy may respond to immunotherapy. However, the symptom management (pain medications, physical therapy) is similar for both.
Should I see a neurologist or my primary care doctor?
Start with your primary care doctor for initial evaluation and blood work. If your symptoms persist or your doctor suspects neuropathy, ask for a referral to a neurologist — ideally one with experience in neuroimmunology or post-infectious neurological conditions. Some academic medical centers have dedicated long COVID clinics with neurological expertise. A neurologist can order specialized testing (skin biopsy, autonomic function testing) that most primary care offices cannot.

